Decoding the Oxygen Reduction Reaction: Mechanistic Insights from Transition Metal Heterostructures
Corresponding Author: Yao Xiao
Nano-Micro Letters,
Vol. 18 (2026), Article Number: 351
Abstract
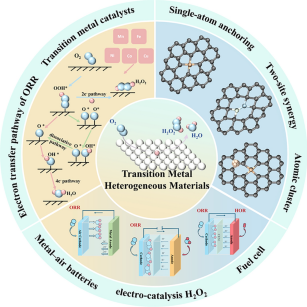
The oxygen reduction reaction (ORR) plays a central role in determining the efficiency and durability of a broad range of electrochemical energy conversion technologies, while its intrinsically slow kinetics and multistep reaction nature remain key challenges. Transition metal-based heterogeneous electrocatalysts, particularly those incorporating Fe, Mn, Co, Ni, and Cu, have been extensively investigated as ORR active systems owing to their earth abundance, diverse coordination environments, and tunable electronic structures. Recent studies show that ORR on these catalysts can diverge into two-electron and four-electron pathways from a common O2 reactant, with pathway selectivity governed by atomic-scale coordination and electronic effects. This review summarizes current understanding of ORR mechanisms on transition metal-based catalysts, with a focus on how atomic-scale structural features are related to reaction activity, selectivity, and stability under different electrolyte conditions. Representative applications in metal–air batteries, fuel cells, and electrochemical hydrogen peroxide production are also discussed to illustrate how mechanistic insights are reflected in practical electrochemical systems. By distilling shared mechanistic features across different systems, this review provides a coherent framework for understanding and guiding ORR catalyst design.
Highlights:
1 This review presents a unified mechanistic framework for oxygen reduction reaction catalysis across diverse transition metal systems (Fe, Mn, Co, Ni, Cu), linking electronic structure, coordination environment, and interfacial effects to pathway selectivity.
2 In situ/operando techniques and interfacial engineering are emphasized as critical tools for atomic-level active site probing and performance optimization.
3 A cross-scale design paradigm bridges molecular-level insights with practical applications in fuel cells, metal-air batteries, and H2O2 electrosynthesis.
Keywords
Download Citation
Endnote/Zotero/Mendeley (RIS)BibTeX
- D.M. Harraz, K.M. Lodaya, B.Y. Tang, Y Surendranath, Homogeneous-heterogeneous bifunctionality in Pd-catalyzed vinyl acetate synthesis. Science 388(6742), eads7913 (2025). https://doi.org/10.1126/science.ads7913
- M. Luo, Z. Zhao, Y. Zhang, Y. Sun, Y. Xing et al., PdMo bimetallene for oxygen reduction catalysis. Nature 574(7776), 81–85 (2019). https://doi.org/10.1038/s41586-019-1603-7
- Y. Yan, S. Xu, H. Li, N.C.S. Selvam, J.Y. Lee et al., Perpendicularly anchored ReSe2 nanoflakes on reduced graphene oxide support for highly efficient hydrogen evolution reactions. Chem. Eng. J. 405, 126728 (2021). https://doi.org/10.1016/j.cej.2020.126728
- R. Chattot, O. Le Bacq, V. Beermann, S. Kühl, J. Herranz et al., Surface distortion as a unifying concept and descriptor in oxygen reduction reaction electrocatalysis. Nat. Mater. 17(9), 827–833 (2018). https://doi.org/10.1038/s41563-018-0133-2
- S. Dey, B. Mondal, S. Chatterjee, A. Rana, S. Amanullah et al., Molecular electrocatalysts for the oxygen reduction reaction. Nat. Rev. Chem. 1(12), 98 (2017). https://doi.org/10.1038/s41570-017-0098
- T. Fukushima, W. Drisdell, J. Yano, Y. Surendranath, Graphite-conjugated pyrazines as molecularly tunable heterogeneous electrocatalysts. J. Am. Chem. Soc. 137(34), 10926–10929 (2015). https://doi.org/10.1021/jacs.5b06737
- A.A. Gewirth, J.A. Varnell, A.M. DiAscro, Nonprecious metal catalysts for oxygen reduction in heterogeneous aqueous systems. Chem. Rev. 118(5), 2313–2339 (2018). https://doi.org/10.1021/acs.chemrev.7b00335
- J. Hong, L. Zhang, Q. Zhu, Z. Du, Y. Zhou et al., A macroporous carbon nanoframe for hosting Mott–Schottky Fe–Co/Mo2C sites as an outstanding bi-functional oxygen electrocatalyst. Mater. Horiz. 10(12), 5969–5982 (2023). https://doi.org/10.1039/D3MH01237A
- J. Wang, M. Sun, X. Zhang, J. Liu, J. He et al., pH-dependent urea electrooxidation: from mechanism to catalysts and applications. Adv. Mater. 38(4), e15043 (2026). https://doi.org/10.1002/adma.202515043
- Z. Hou, C. Cui, Y. Yang, Z. Huang, Y. Zhuang et al., Strong metal-support interactions in heterogeneous oxygen electrocatalysis. Small 20(52), 2407167 (2024). https://doi.org/10.1002/smll.202407167
- C. Hu, G. Xing, W. Han, Y. Hao, C. Zhang et al., Inhibiting demetalation of Fe-N-C via Mn sites for efficient oxygen reduction reaction in zinc-air batteries. Adv. Mater. 36(32), 2405763 (2024). https://doi.org/10.1002/adma.202405763
- J.J. Huang, Y. Yang, D. Weinstock, C.R. Bundschu, Q. Li et al., Multimodal in situ X-ray mechanistic studies of a bimetallic oxide electrocatalyst in alkaline media. Nat. Catal. 8(2), 116–125 (2025). https://doi.org/10.1038/s41929-025-01289-7
- B. Ji, J. Gou, Y. Zheng, X. Pu, Y. Wang et al., Coordination chemistry of large-sized yttrium single-atom catalysts for oxygen reduction reaction. Adv. Mater. 35(24), 2300381 (2023). https://doi.org/10.1002/adma.202300381
- N. Kuwamura, T. Konno, Heterometallic coordination polymers as heterogeneous electrocatalysts. Inorg. Chem. Front. 8(10), 2634–2649 (2021). https://doi.org/10.1039/d1qi00112d
- Y. Li, H.-R. Wu, Y. Yu, M.-Y. Chen, K.-M. Zhao et al., High-shell sulfur doping enhances Mn-N4 spin states and boosts oxygen reduction reaction performance in both acidic and alkaline media. Small 21(11), 2411678 (2025). https://doi.org/10.1002/smll.202411678
- Y. Pei, D.P. Wilkinson, E. Gyenge, Insights into the electrochemical behavior of manganese oxides as catalysts for the oxygen reduction and evolution reactions: monometallic core-shell Mn/Mn3O4. Small 19(19), 2204585 (2023). https://doi.org/10.1002/smll.202204585
- S.C. Perry, D. Pangotra, L. Vieira, L.-I. Csepei, V. Sieber et al., Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3(7), 442–458 (2019). https://doi.org/10.1038/s41570-019-0110-6
- F.N.I. Sari, Y.-C. Lai, Y.-J. Huang, X.-Y. Wei, H. Pourzolfaghar et al., Electronic structure engineering in NiFe sulfide via a third metal doping as efficient bifunctional OER/ORR electrocatalyst for rechargeable zinc-air battery. Adv. Funct. Mater. 34(21), 2310181 (2024). https://doi.org/10.1002/adfm.202310181
- T. Sharifi, E. Gracia-Espino, A. Chen, G. Hu, T. Wågberg, Oxygen reduction reactions on single-or few-atom discrete active sites for heterogeneous catalysis. Adv. Energy Mater. 10(11), 1902084 (2020). https://doi.org/10.1002/aenm.201902084
- J. Song, Z.-X. Qian, J. Yang, X.-M. Lin, Q. Xu et al., In situ/operando investigation for heterogeneous electro-catalysts: from model catalysts to state-of-the-art catalysts. ACS Energy Lett. 9(9), 4414–4440 (2024). https://doi.org/10.1021/acsenergylett.4c01488
- D.-W. Wang, D. Su, Heterogeneous nanocarbon materials for oxygen reduction reaction. Energy Environ. Sci. 7(2), 576 (2014). https://doi.org/10.1039/c3ee43463j
- X. Wang, N. Zhang, H. Shang, H. Duan, Z. Sun et al., Precisely designing asymmetrical selenium-based dual-atom sites for efficient oxygen reduction. Nat. Commun. 16(1), 470 (2025). https://doi.org/10.1038/s41467-025-55862-6
- H. Tian, A. Song, H. Tian, J. Liu, G. Shao et al., Single-atom catalysts for high-energy rechargeable batteries. Chem. Sci. 12(22), 7656–7676 (2021). https://doi.org/10.1039/d1sc00716e
- Y. Wang, G.I.N. Waterhouse, L. Shang, T. Zhang, Electrocatalytic oxygen reduction to hydrogen peroxide: from homogeneous to heterogeneous electrocatalysis. Adv. Energy Mater. 11(15), 2003323 (2021). https://doi.org/10.1002/aenm.202003323
- L.-H. Xu, Q. Wang, L. Hu, D. Shen, S. Chu et al., Engineering asymmetric bimetallic CoM (M = Ni, Fe, Mn, Cu) nanops encapsulated in freestanding wood-derived carbon electrodes for enhanced ORR kinetics in zinc-air batteries. Small 21(5), e2410290 (2025). https://doi.org/10.1002/smll.202410290
- Y. Yan, R. Yu, M. Liu, Z. Qu, J. Yang et al., General synthesis of neighboring dual-atomic sites with a specific pre-designed distance via an interfacial-fixing strategy. Nat. Commun. 16(1), 334 (2025). https://doi.org/10.1038/s41467-024-55630-y
- H. Yang, N. An, Z. Kang, P.W. Menezes, Z. Chen, Understanding advanced transition metal-based two electron oxygen reduction electrocatalysts from the perspective of phase engineering. Adv. Mater. 36(25), 2400140 (2024). https://doi.org/10.1002/adma.202400140
- S. Yang, X. Liu, S. Li, W. Yuan, L. Yang et al., The mechanism of water oxidation using transition metal-based heterogeneous electrocatalysts. Chem. Soc. Rev. 53(11), 5593–5625 (2024). https://doi.org/10.1039/d3cs01031g
- Z. Jiang, X. Yang, J. Zhang, J. Yang, B. Sun et al., From conventional two-electron to emerging multi-electron zinc-iodine batteries: advantages, challenges, and future perspectives. Adv. Funct. Mater. 35(50), e11754 (2025). https://doi.org/10.1002/adfm.202511754
- H. Zhang, H.-C. Chen, S. Feizpoor, L. Li, X. Zhang et al., Tailoring oxygen reduction reaction kinetics of Fe-N-C catalyst via spin manipulation for efficient zinc-air batteries. Adv. Mater. 36(25), e2400523 (2024). https://doi.org/10.1002/adma.202400523
- H. Tian, Y. Lei, B. Sun, C.-C. Yang, C.-L. Chen et al.,P-d orbital hybridization induced by transition metal atom sites for room temperature sodium sulfur batteries. Natl. Sci. Rev. 12(7), nwaf241 (2025). https://doi.org/10.1093/nsr/nwaf241
- Z. Ma, A. Song, Z. Liu, Y. Guo, X. Yang et al., Nanoconfined expansion behavior of hollow MnS@Carbon anode with extended lithiation cyclic stability. Adv. Funct. Mater. 33(28), 2301112 (2023). https://doi.org/10.1002/adfm.202301112
- S. Zhao, Y. Yang, Z. Tang, Insight into structural evolution, active sites, and stability of heterogeneous electrocatalysts. Angew. Chem. Int. Ed. 61(11), e202110186 (2022). https://doi.org/10.1002/anie.202110186
- X. Zhao, Z.H. Levell, S. Yu, Y. Liu, Atomistic understanding of two-dimensional electrocatalysts from first principles. Chem. Rev. 122(12), 10675–10709 (2022). https://doi.org/10.1021/acs.chemrev.1c00981
- J. Zheng, D. Meng, J. Guo, X. Liu, L. Zhou et al., Defect engineering for enhanced electrocatalytic oxygen reaction on transition metal oxides: the role of metal defects. Adv. Mater. 36(28), 2405129 (2024). https://doi.org/10.1002/adma.202405129
- H.-X. Zhong, Y. Zhang, X.-B. Zhang, Superior oxygen reduction electrocatalyst: hollow porous spinel microsphere. Chem 4(2), 196–198 (2018). https://doi.org/10.1016/j.chempr.2018.01.015
- B. Zhou, N. Xu, T. Lu, Y. Wang, S. Lou et al., Dual-carbon assisted oxygen vacancy engineering for optimizing Mn(III) sites to enhance Zn–air battery performances. Adv. Funct. Mater. 35(4), 2414269 (2025). https://doi.org/10.1002/adfm.202414269
- H. Zhou, H. Liu, Y. Sun, L. Jiang, J. Xiao et al., Synergistic effect of single atomic Ce sites and CeO2 nanops for boosting oxygen reduction reaction. Adv. Energy Mater. 15(14), 2404689 (2025). https://doi.org/10.1002/aenm.202404689
- M. Zhou, J. Guo, R. Lu, J. Li, S. Lee et al., Amorphous metal metaphosphate for oxygen reduction. Interdiscip. Mater. 4(2), 309–320 (2025). https://doi.org/10.1002/idm2.12228
- X. Zou, Q. Lu, J. Wu, K. Zhang, M. Tang et al., Screening spinel oxide supports for RuO2 to boost bifunctional electrocatalysts for advanced Zn–air batteries. Adv. Funct. Mater. 34(36), 2401134 (2024). https://doi.org/10.1002/adfm.202401134
- G. Gao, Z. Sun, X. Chen, Z. Guang, B. Sun et al., Recent advances in hydrogen production coupled with alternative oxidation reactions. Coord. Chem. Rev. 509, 215777 (2024). https://doi.org/10.1016/j.ccr.2024.215777
- G. Gao, C. Xiao, R. Zhang, W. Chen, C. Liu et al., ZIF-67 derivatives in electrocatalysis. Coord. Chem. Rev. 523, 216296 (2025). https://doi.org/10.1016/j.ccr.2024.216296
- G. Gao, G. Zhao, G. Zhu, B. Sun, Z. Sun et al., Recent advancements in noble-metal electrocatalysts for alkaline hydrogen evolution reaction. Chin. Chemical Lett. 36(1), 109557 (2025). https://doi.org/10.1016/j.cclet.2024.109557
- X. Zhang, J. Lu, J. Liu, M. Sun, G. Zhang et al., Curvature geometry-spin electronics-catalytic dynamics coupling in emerging catalytic engineering. Chem. Soc. Rev. 55(5), 2691–2730 (2026). https://doi.org/10.1039/d5cs01114k
- Q. Huang, F. Wang, Z. Sun, B. Zhang, W. Li et al., In situ growth and dynamic transformation of nickel chelate nanoarrays into reactive surface reconstituted heterostructure for overall water splitting. Adv. Funct. Mater. 34(46), 2407407 (2024). https://doi.org/10.1002/adfm.202407407
- L. Shang, Y. Ni, Y. Wang, W. Yang, L. Wang et al., Single-nanometer spinel with precise cation distribution for enhanced oxygen reduction. Adv. Mater. 36(50), e2413141 (2024). https://doi.org/10.1002/adma.202413141
- H. Tian, H. Tian, S. Wang, S. Chen, F. Zhang et al., High-power lithium-selenium batteries enabled by atomic cobalt electrocatalyst in hollow carbon cathode. Nat. Commun. 11(1), 5025 (2020). https://doi.org/10.1038/s41467-020-18820-y
- X. Zhang, Z. Song, B. Liu, B. Liu, J. Liu et al., The interfacial charge change enhanced by Pr0.6 Sm0.4 Co0.8 Mn0.2O3 activated peroxymonosulfate was used for the efficient degradation of tetracycline under the nanoscale domain limiting and distance effect. Mater. Today Sustain. 28, 101044 (2024). https://doi.org/10.1016/j.mtsust.2024.101044
- X. Zhang, Z. Song, B. Liu, J. Liu, Y. Huang et al., Ni/Fe bimetallic atom cluster activate PMS to promote 1O2 production to efficiently remove BPA. ChemistrySelect 9(8), e202303088 (2024). https://doi.org/10.1002/slct.202303088
- Y. Yan, M. Wu, L. Zhou, W. Chen, L. Han et al., Enhancing electrocatalytic activity through targeted local electrolyte micro-environment. Adv. Funct. Mater. 35(19), 2419328 (2025). https://doi.org/10.1002/adfm.202419328
- S. Kong, J. Liu, X. Zhang, J. He, G. Zhang et al., Bridging organic and inorganic domains: advances and applications of hybrid materials in electrocatalysis. Adv. Energy Mater. 16(5), e05010 (2026). https://doi.org/10.1002/aenm.202505010
- Z. Song, X. Zhang, B. Liu, J. Liu, Y. Huang et al., Highly active Ni atomic clusters loaded with coal gasification slag derivatives effectively remove tetracycline by activating persulfate to enhance electron transfer ability. J. Water Process Eng. 66, 105982 (2024). https://doi.org/10.1016/j.jwpe.2024.105982
- X. Shi, Y. Jiang, B. Zeng, Z. Sun, M. Yun et al., In situ electrochemical production of solid peroxide from urine. Nat. Catal. 8(1), 67–78 (2025). https://doi.org/10.1038/s41929-024-01277-3
- J. Liu, Z. Song, B. Liu, X. Zhang, Y. Huang et al., Bimetallic Ni/Fe atom cluster catalysts enhance non-free radical degradation of organic pollutant phenol. Catal. Lett. 154(5), 2182–2196 (2024). https://doi.org/10.1007/s10562-023-04472-2
- J. Liu, Y. Huang, X. Song, Z. Song, X. Zhang et al., Ni-Co synergistic regulation of catalyst surface charge density for efficient DNA base degradation in water. J. Water Process. Eng. 71, 107313 (2025). https://doi.org/10.1016/j.jwpe.2025.107313
- G. Zhang, X. Zhang, J. Liu, J. He, W. Ge et al., Transition metal-based electrocatalysts for CO2 reduction towards ethanol. Coord. Chem. Rev. 550, 217403 (2026). https://doi.org/10.1016/j.ccr.2025.217403
- C. Li, Y. Yang, J. Lu, L. Ren, X. Zhang et al., Micro-Cu doped Co3O4 as an effective oxygen reduction nano-flower-like catalyst to enhance the power output of air cathode microbial fuel cell. Catal. Lett. 154(11), 6080–6093 (2024). https://doi.org/10.1007/s10562-024-04779-8
- Y. Huang, Z. Song, B. Liu, X. Zhang, J. Liu et al., Highly active Co is injected into the PrSmMnO3 parent structure to promote the 1O2 pathway to efficiently degrade residual chloroquine phosphate in wastewater. Catal. Lett. 155(1), 3 (2024). https://doi.org/10.1007/s10562-024-04844-2
- M. Du, B. Chu, Q. Wang, C. Li, Y. Lu et al., Dual Fe/I single-atom electrocatalyst for high-performance oxygen reduction and wide-temperature quasi-solid-state Zn-air batteries. Adv. Mater. 36(47), 2412978 (2024). https://doi.org/10.1002/adma.202412978
- J. Zhang, Y. Mou, W. Suo, S. Yang, J. Shen et al., Single-atomic Co-N-C sites anchored on helical carbonaceous nanotubes for the oxygen reduction reaction. Adv. Funct. Mater. 35(12), 2417621 (2025). https://doi.org/10.1002/adfm.202417621
- C. Brea, G. Hu, Dual-atom catalysts for the oxygen reduction reaction: unraveling atomic structures under reaction conditions. J. Am. Chem. Soc. 147(22), 19210–19216 (2025). https://doi.org/10.1021/jacs.5c04776
- Y. Cao, Y. Liu, X. Zheng, J. Yang, H. Wang et al., Quantifying asymmetric coordination to correlate with oxygen reduction activity in Fe-based single-atom catalysts. Angew. Chem. Int. Ed. 64(14), e202423556 (2025). https://doi.org/10.1002/anie.202423556
- X. Cui, R. Jin, L. Gao, M. Wu, Y. Liu et al., High-loading single atoms via hierarchically porous nanospheres for oxygen reduction reaction with superior activity and durability. Adv. Funct. Mater. 35(39), 2510108 (2025). https://doi.org/10.1002/adfm.202510108
- M. Dan, X. Zhang, C. Du, Z. Guo, J. Zhang et al., Atomically dispersed Fe confined into MnO nanoclusters enhances alkaline oxygen reduction activity and stability. Angew. Chem. Int. Ed. 64(29), e202501531 (2025). https://doi.org/10.1002/anie.202501531
- M. Deng, D. Wang, Y. Li, General design concept of high-performance single-atom-site catalysts for H2O2 electrosynthesis. Adv. Mater. 36(24), e2314340 (2024). https://doi.org/10.1002/adma.202314340
- H. Huang, M. Sun, K. Chen, Y. Che, X. Tang et al., Unlocking the potential of Mn-based catalyst for durable two-electron oxygen reduction in acid at high current densities. Angew. Chem. Int. Ed. 64(33), e202511844 (2025). https://doi.org/10.1002/anie.202511844
- S.-M. Jung, S. Kim, J. An, K.-S. Kim, M. Kim et al., Synergistic effects of Co–N4 and Ni–N4 sites in 2D conductive metal–organic framework electrocatalysts for enhanced oxygen reduction reaction performance. ACS Catal. 15(7), 5568–5576 (2025). https://doi.org/10.1021/acscatal.4c05773
- S. Lin, J. Wang, J. Chen, P. Lin, H. Wang et al., Electrochemical pilot H2O2 production by solid-state electrolyte reactor: insights from a hybrid catalyst for 2-electron oxygen reduction reaction. Angew. Chem. Int. Ed. 64(19), e202502144 (2025). https://doi.org/10.1002/anie.202502144
- C. Liu, W. Shi, Y. Yuan, K. Zhu, Q. Zhang et al., Cascade hydrogen peroxide reduction reaction endows Cu-Fe dual-atom catalyst with durable oxygen reduction performance. Adv. Funct. Mater. 35(35), 2503079 (2025). https://doi.org/10.1002/adfm.202503079
- C. Liu, R. Yang, J. Wang, B. Liu, X. Chang et al., Synergistic catalysts with Fe single atoms and Fe3C clusters for accelerated oxygen adsorption kinetics in oxygen reduction reaction. Angew. Chem. Int. Ed. 64(21), e202501266 (2025). https://doi.org/10.1002/anie.202501266
- L. Liu, F. Chen, H. Yang, X. Yan, J. Ren et al., Asymmetric coordination strategy of Cu single-atom catalyst for robust all-pH oxygen reduction reaction. Small 21(32), e2503745 (2025). https://doi.org/10.1002/smll.202503745
- M. Liu, Y. Li, L. Yang, P. Zhao, J. Li et al., Defect-triggered orbital hybridization in FeMn dual-atom catalysts toward Sabatier-optimized oxygen reduction. Angew. Chem. Int. Ed. 64(28), e202505268 (2025). https://doi.org/10.1002/anie.202505268
- S. Liu, Q. Meyer, D. Xu, Y. Cheng, L. Osmieri et al., Breaking the activity and stability trade-off of platinum-free catalysts for the oxygen reduction reaction in hydrogen fuel cells. ACS Nano 19(21), 19524–19551 (2025). https://doi.org/10.1021/acsnano.5c03610
- Y. Liu, Y. Yang, X. Lin, Y. Lin, Z. Zhuo et al., The geometric-electronic coupled design of diatomic catalyst towards oxygen reduction reaction. Nat. Commun. 16(1), 5158 (2025). https://doi.org/10.1038/s41467-025-60170-0
- X. Lu, F. Xiang, S. Li, W. Liu, Y. Zeng et al., Hydrogen-bond-assisted synthesis of single-atom and nanocluster synergistic sites for enhanced oxygen reduction reaction. Adv. Funct. Mater. 35(40), 2506982 (2025). https://doi.org/10.1002/adfm.202506982
- Q. Ma, Y. Liao, Q. Zhao, R. Gan, Y. Ran et al., Triggering synergistic electronic effect via electron-directed transfer within PtNPs-Fe/NC oxygen reduction catalyst for zinc-air batteries. Small 21(14), 2500344 (2025). https://doi.org/10.1002/smll.202500344
- D. Shen, F. Sun, Z. Liang, H. Fu, L. Wang, Axial-N induced square-pyramidal crystal filed of atomically iron sites for enhancing acidic oxygen reduction. Angew. Chem. Int. Ed. 64(33), e202505937 (2025). https://doi.org/10.1002/anie.202505937
- Z. Wan, Z. Ma, X. Deng, Y. Wu, J. Li et al., Weakening the dissociation barrier of hydroxyl in Fe–N–C catalysts via precisely manipulating d–p orbital hybridization behaviors for efficient oxygen reduction reaction. Adv. Energy Mater. 15(31), 2501630 (2025). https://doi.org/10.1002/aenm.202501630
- P. Wang, Y. Xu, Y. Li, P. Xie, H. Li et al., Engineering active CeO2/Fe3C interfacial sites to generate high-charge-density Fe for enhanced oxygen reduction reaction efficiency. Adv. Funct. Mater. 35(43), 2503577 (2025). https://doi.org/10.1002/adfm.202503577
- H. Tian, A. Song, P. Zhang, K. Sun, J. Wang et al., High durability of Fe–N–C single-atom catalysts with carbon vacancies toward the oxygen reduction reaction in alkaline media. Adv. Mater. 35(14), 2210714 (2023). https://doi.org/10.1002/adma.202210714
- B. Zhang, J. Dang, H. Li, J.-J. Wang, D. Xu et al., Orderly stacked “tile” architecture with single-atom iron boosts oxygen reduction in liquid and solid-state Zn–air batteries. Adv. Funct. Mater. 35(34), 2502834 (2025). https://doi.org/10.1002/adfm.202502834
- T. Zhang, W. Wang, W. Liu, Z. Guo, J. Liu, Residual ligand-functionalized ultrathin Ni(OH)2 via reconstruction for high-rate HO2− electrosynthesis. Nat. Commun. 16(1), 5240 (2025). https://doi.org/10.1038/s41467-025-60467-0
- Z. Zhang, Z. Zheng, N. Ma, E. Picheau, N. Sakai et al., Composition tuning and heterostructure construction of Fe-doped Co-Ni hydroxide nanosheets for boosting oxygen electrocatalysis in rechargeable Zn-air batteries. Chem. Eng. J. 509, 161248 (2025). https://doi.org/10.1016/j.cej.2025.161248
- Y. Zhao, Z. Gao, S. Zhang, X. Guan, W. Xu et al., Asymmetric-charge-distributed Co-Mn diatomic catalyst enables efficient oxygen reduction reaction. Adv. Funct. Mater. 35(37), 2504260 (2025). https://doi.org/10.1002/adfm.202504260
- J. Zou, L. Bao, Q. Sun, C. Bao, H. Chen et al., Oxygen reduction reaction catalysts for zinc-air batteries featuring single cobalt atoms in a nitrogen-doped 3D-interconnected porous graphene framework. Small 21(8), 2409506 (2025). https://doi.org/10.1002/smll.202409506
- C. Brea, G. Hu, Mechanistic insight into dual-metal-site catalysts for the oxygen reduction reaction. ACS Catal. 13(7), 4992–4999 (2023). https://doi.org/10.1021/acscatal.3c00090
- X. Cao, H. Guo, Y. Han, M. Li, C. Shang et al., Sandwiching intermetallic Pt3Fe and ionomer with porous N-doped carbon layers for oxygen reduction reaction. Nat. Commun. 16(1), 2851 (2025). https://doi.org/10.1038/s41467-025-58116-7
- T. He, Y. Chen, Q. Liu, B. Lu, X. Song et al., Theory-guided regulation of FeN4 spin state by neighboring Cu atoms for enhanced oxygen reduction electrocatalysis in flexible metal–air batteries. Angew. Chem. Int. Ed. 61(27), e202201007 (2022). https://doi.org/10.1002/anie.202201007
- J. Huang, C. Yu, J. Li, W. Xiao, J.B. Zhong et al., Rare-earth lanthanum-nitrogen-carbon enhanced by abundant microspores for efficient oxygen reduction reaction. J. Energy Chem. 106, 812–822 (2025). https://doi.org/10.1016/j.jechem.2025.02.021
- M. Tong, F. Sun, Y. Xie, Y. Wang, Y. Yang et al., Operando cooperated catalytic mechanism of atomically dispersed Cu−N4 and Zn−N4 for promoting oxygen reduction reaction. Angew. Chem. Int. Ed. 60(25), 14005–14012 (2021). https://doi.org/10.1002/anie.202102053
- M. Zhang, H. Li, J. Chen, F.-X. Ma, L. Zhen et al., Transition metal (Co, Ni, Fe, Cu) single-atom catalysts anchored on 3D nitrogen-doped porous carbon nanosheets as efficient oxygen reduction electrocatalysts for Zn-air battery. Small 18(34), e2202476 (2022). https://doi.org/10.1002/smll.202202476
- L. Zong, K. Fan, P. Li, F. Lu, B. Li et al., Promoting oxygen reduction reaction on atomically dispersed Fe sites via establishing hydrogen bonding with the neighboring P atoms. Adv. Energy Mater. 13(5), 2203611 (2023). https://doi.org/10.1002/aenm.202203611
- R. Jinnouchi, K. Kodama, T. Hatanaka, Y. Morimoto, First principles based mean field model for oxygen reduction reaction. Phys. Chem. Chem. Phys. 13(47), 21070 (2011). https://doi.org/10.1039/c1cp21349k
- V. Viswanathan, H.A. Hansen, J. Rossmeisl, J.K. Nørskov, Unifying the 2e− and 4e− reduction of oxygen on metal surfaces. J. Phys. Chem. Lett. 3(20), 2948–2951 (2012). https://doi.org/10.1021/jz301476w
- H. Chen, Z. Gao, N.T. Nguyen, Z. Li, R.-T. Gao et al., Bias-free photoelectrochemical water oxidation coupled with electrochemical oxygen reduction reaction via Fe-based electrodes with long-term operation. Adv. Funct. Mater. 35(14), 2418670 (2025). https://doi.org/10.1002/adfm.202418670
- H. Liu, J. Huang, K. Feng, R. Xiong, S. Ma et al., Reconstructing the coordination environment of Fe/Co dual-atom sites towards efficient oxygen electrocatalysis for Zn–air batteries. Angew. Chem. Int. Ed. 64(7), e202419595 (2025). https://doi.org/10.1002/anie.202419595
- S. Bhattacharyya, D. Samanta, S. Roy, V.P. Haveri Radhakantha, T.K. Maji, In situ stabilization of Au and co nanops in a redox-active conjugated microporous polymer matrix: facile heterogeneous catalysis and electrocatalytic oxygen reduction reaction activity. ACS Appl. Mater. Interfaces 11(5), 5455–5461 (2019). https://doi.org/10.1021/acsami.8b20610
- V. Beermann, M. Gocyla, E. Willinger, S. Rudi, M. Heggen et al., Rh-doped Pt–Ni octahedral nanops: understanding the correlation between elemental distribution, oxygen reduction reaction, and shape stability. Nano Lett. 16(3), 1719–1725 (2016). https://doi.org/10.1021/acs.nanolett.5b04636
- Y. Chen, J. Xu, P. He, Y. Qiao, S. Guo et al., Metal-air batteries: progress and perspective. Sci. Bull. 67(23), 2449–2486 (2022). https://doi.org/10.1016/j.scib.2022.11.027
- J. Sun, T. Tang, S. Zhang, S. Chen, Y. Duan et al., A dual-atom La2 catalyst for the oxygen reduction reaction. Angew. Chem. Int. Ed. 64(34), e202509063 (2025). https://doi.org/10.1002/anie.202509063
- Y. Wang, J. Wu, Q. Zhang, Y. Tan, J. Gao et al., Defect-induced Zn–Co pair active site for high-efficiency electrosynthesis of H2O2. Matter 8(12), 102479 (2025). https://doi.org/10.1016/j.matt.2025.102479
- P.H. van Langevelde, K. Ležaić, J.F.J. Coelho, D.G.H. Hetterscheid, F. De Bon, Interplay between the oxygen reduction reaction and atom transfer radical polymerization with molecular Cu-based catalysts in water. ACS Catal. 15(16), 14548–14563 (2025). https://doi.org/10.1021/acscatal.5c04928
- Y. Zhao, J. Wan, C. Ling, Y. Wang, H. He et al., Acidic oxygen reduction by single-atom Fe catalysts on curved supports. Nature 644(8077), 668–675 (2025). https://doi.org/10.1038/s41586-025-09364-6
- M. Sun, J. Chen, Z. Zhang, Y. Jing, M. Zhao et al., Ferromagnetic ordering outperforms coordination effects in governing oxygen reduction catalysis on high-index nickel single crystals. Angew. Chem. Int. Ed. 64(31), e202504869 (2025). https://doi.org/10.1002/anie.202504869
- Z. Lu, Z. Wang, Z. Yang, X. Jin, L. Tong et al., Engineering CoN4 and FeN4 dual sites with adjacent nanoclusters on flexible porous carbon fibers for enhanced electrocatalytic oxygen reduction and evolution. Adv. Funct. Mater. 35(16), 2418489 (2025). https://doi.org/10.1002/adfm.202418489
- M.L. Rigsby, D.J. Wasylenko, M.L. Pegis, J.M. Mayer, Medium effects are as important as catalyst design for selectivity in electrocatalytic oxygen reduction by iron–porphyrin complexes. J. Am. Chem. Soc. 137(13), 4296–4299 (2015). https://doi.org/10.1021/jacs.5b00359
- H. Wang, L. Cao, Y. Feng, J. Chen, W. Feng et al., Facile synthesis of defect-rich Fe-N-C hybrid from fullerene/ferrotetraphenylporphyrin as efficient oxygen reduction electrocatalyst for Zn-air battery. Chin. Chem. Lett. 34(5), 107601 (2023). https://doi.org/10.1016/j.cclet.2022.06.024
- Z. Wang, R. Xu, Q. Ye, X. Jin, Z. Lu et al., Tailoring first coordination sphere of dual-metal atom sites boosts oxygen reduction and evolution activities. Adv. Funct. Mater. 34(28), 2315376 (2024). https://doi.org/10.1002/adfm.202315376
- L. Wu, Y. Chen, C. Shao, L. Wang, B. Li, Engineering synergetic Fe-co atomic pairs anchored on porous carbon for enhanced oxygen reduction reaction. Adv. Funct. Mater. 34(48), 2408257 (2024). https://doi.org/10.1002/adfm.202408257
- P.-F. Xie, H. Zhong, L. Fang, Z. Lyu, W.-J. Yu et al., Molecular Fe-N4 moieties coupled with atomic Co-N4 sites toward improved oxygen reduction performance. Adv. Funct. Mater. 34(32), 2314554 (2024). https://doi.org/10.1002/adfm.202314554
- D. Xue, S. Zhao, B.-A. Lu, Y. Yu, Y. Wei et al., Disentangling the activity-stability trade-off of pyrrolic N-coordinated Fe-N4 catalytic sites for long-life oxygen reduction reaction in acidic medium. Adv. Energy Mater. 14(12), 2303733 (2024). https://doi.org/10.1002/aenm.202303733
- H. Yang, H. Wang, S. Ji, V. Linkov, R. Wang, Synergy between isolated-Fe3O4 nanops and CNx layers derived from lysine to improve the catalytic activity for oxygen reduction reaction. Int. J. Hydrog. Energy 39(8), 3739–3745 (2014). https://doi.org/10.1016/j.ijhydene.2013.12.160
- S. Yin, H. Yi, M. Liu, J. Yang, S. Yang et al., An in situ exploration of how Fe/N/C oxygen reduction catalysts evolve during synthesis under pyrolytic conditions. Nat. Commun. 15, 6229 (2024). https://doi.org/10.1038/s41467-024-50629-x
- P. Zhang, H.-C. Chen, H. Zhu, K. Chen, T. Li et al., Inter-site structural heterogeneity induction of single atom Fe catalysts for robust oxygen reduction. Nat. Commun. 15(1), 2062 (2024). https://doi.org/10.1038/s41467-024-46389-3
- S. Ji, Y. Wang, H. Liu, X. Lu, C. Guo et al., Regulating the electronic synergy of asymmetric atomic Fe sites with adjacent defects for boosting activity and durability toward oxygen reduction. Adv. Funct. Mater. 34(29), 2314621 (2024). https://doi.org/10.1002/adfm.202314621
- P. Rao, Y. Liu, X. Shi, Y. Yu, Y. Zhou et al., Protection of Fe single-atoms by Fe clusters for chlorine-resistant oxygen reduction reaction. Adv. Funct. Mater. 34(46), 2407121 (2024). https://doi.org/10.1002/adfm.202407121
- J. Liu, Y. Liu, B. Nan, D. Wang, C. Allen et al., A two-in-one strategy to simultaneously boost the site density and turnover frequency of Fe−N−C oxygen reduction catalysts. Angew. Chem. Int. Ed. 64(14), e202425196 (2025). https://doi.org/10.1002/anie.202425196
- J. Qiao, C. Lu, L. Kong, J. Zhang, Q. Lin et al., Spin engineering of Fe-N-C by axial ligand modulation for enhanced bifunctional oxygen catalysis. Adv. Funct. Mater. 34(51), 2409794 (2024). https://doi.org/10.1002/adfm.202409794
- R. Sui, B. Liu, C. Chen, X. Tan, C. He et al., Constructing asymmetric Fe-Nb diatomic sites to enhance ORR activity and durability. J. Am. Chem. Soc. 146(38), 26442–26453 (2024). https://doi.org/10.1021/jacs.4c09642
- G. Yang, H. Cai, N. Zhang, B. Wang, C. Liang et al., Regulation of d-orbital electron in Fe-N4 by high-entropy atomic clusters for highly active and durable oxygen reduction reaction. Adv. Funct. Mater. 34(46), 2407775 (2024). https://doi.org/10.1002/adfm.202407775
- M. Yuan, Y. Liu, Y. Du, Z. Xiao, H. Li et al., Dual-shelled hollow leafy carbon support with atomically dispersed (N, S)-bridged hydroxy-coordinated asymmetric Fe sites for oxygen reduction. Adv. Funct. Mater. 34(29), 2401484 (2024). https://doi.org/10.1002/adfm.202401484
- Y. Zhu, B. Zhang, X. Liu, D.-W. Wang, D.S. Su, Unravelling the structure of electrocatalytically active Fe–N complexes in carbon for the oxygen reduction reaction. Angew. Chem. Int. Ed. 53(40), 10673–10677 (2014). https://doi.org/10.1002/anie.201405314
- X. Lin, D. Liu, L. Shi, F. Liu, F. Ye et al., Second-shell coordination environment modulation for MnN4 active sites by oxygen doping to boost oxygen reduction performance. Small 2407146 (2024). https://doi.org/10.1002/smll.202407146
- Y. Qin, C. Guo, Z. Ou, C. Xu, Q. Lan et al., Regulating single-atom Mn sites by precisely axial pyridinic-nitrogen coordination to stabilize the oxygen reduction. J. Energy Chem. 80, 542–552 (2023). https://doi.org/10.1016/j.jechem.2023.01.048
- K. Kim, G. Kim, T. Jeong, W. Lee, Y. Yang et al., Activating the Mn single atomic center for an efficient actual active site of the oxygen reduction reaction by spin-state regulation. J. Am. Chem. Soc. 146(49), 34033–34042 (2024). https://doi.org/10.1021/jacs.4c13137
- Y. Song, C. Han, W. Li, X. Yi, Q. Liao et al., Engineering bimetallic cluster architectures: Harnessing unique “remote synergy effect” between Mn and Y for enhanced electrocatalytic oxygen reduction reaction. eScience 5(3), 100332 (2025). https://doi.org/10.1016/j.esci.2024.100332
- T. Stracensky, L. Jiao, Q. Sun, E. Liu, F. Yang et al., Bypassing formation of oxide intermediate via chemical vapor deposition for the synthesis of an Mn-N-C catalyst with improved ORR activity. ACS Catal. 13(22), 14782–14791 (2023). https://doi.org/10.1021/acscatal.3c01982
- Z. Luo, J. Xie, J. Cheng, F. Wei, S. Lyu et al., Spin-state manipulation of atomic manganese center by phosphide-support interactions for enhanced oxygen reduction. Adv. Mater. 37(29), 2504585 (2025). https://doi.org/10.1002/adma.202504585
- Y. Li, Z. Li, K. Shi, L. Luo, H. Jiang et al., Single-atom Mn catalysts via integration with Mn sub nano-clusters synergistically enhance oxygen reduction reaction. Small 20(22), 2309727 (2024). https://doi.org/10.1002/smll.202309727
- H. Tian, L. Zeng, Y. Huang, Z. Ma, G. Meng et al., In situ electrochemical Mn(III)/Mn(IV) generation of Mn(II)O electrocatalysts for high-performance oxygen reduction. Nano-Micro Lett. 12(1), 161 (2020). https://doi.org/10.1007/s40820-020-00500-7
- S. Ye, D. Zhang, Z. Ou, L. Zheng, W. Liu et al., Electron donor–acceptor activated single atomic sites for boosting oxygen reduction reaction. Adv. Funct. Mater. 34(40), 2405884 (2024). https://doi.org/10.1002/adfm.202405884
- C. Zhao, H. Li, W. Wang, J. Xu, B. Shao et al., Revealing the critical role of the lone pair electrons of Bi3+ in electrocatalytic oxygen reduction reaction on Mn-based BiMn2O5 mullite surface. Adv. Funct. Mater. 35(9), 2416257 (2025). https://doi.org/10.1002/adfm.202416257
- Y. Jing, Y. Cheng, L. Wang, Y. Liu, B. Yu et al., MOF-derived Co, Fe, and Ni Co-doped N-enriched hollow carbon as efficient electrocatalyst for oxygen reduction reaction. Chem. Eng. J. 397, 125539 (2020). https://doi.org/10.1016/j.cej.2020.125539
- B. Li, H. Nam, J. Zhao, J. Chang, N. Lingappan et al., Nanoreactor of nickel-containing carbon-shells as oxygen reduction catalyst. Adv. Mater. 29(7), 1605083 (2017). https://doi.org/10.1002/adma.201605083
- H. Li, S. Dai, Y. Wu, Q. Dong, J. Chen et al., Atomic scaled depth correlation to the oxygen reduction reaction performance of single atom Ni alloy to the NiO2 supported Pd nanocrystal. Adv. Sci. 10(11), 2207109 (2023). https://doi.org/10.1002/advs.202207109
- M. Li, H. Zhu, Q. Yuan, T. Li, M. Wang et al., Proximity electronic effect of Ni/Co diatomic sites for synergistic promotion of electrocatalytic oxygen reduction and hydrogen evolution. Adv. Funct. Mater. 33(4), 2210867 (2023). https://doi.org/10.1002/adfm.202210867
- Y. Sun, K. Fan, J. Li, L. Wang, Y. Yang et al., Boosting electrochemical oxygen reduction to hydrogen peroxide coupled with organic oxidation. Nat. Commun. 15(1), 6098 (2024). https://doi.org/10.1038/s41467-024-50446-2
- Q. Zhang, Z. Zheng, R. Gao, X. Xiao, M. Jiao et al., Constructing bipolar dual-active sites through high-entropy-induced electric dipole transition for decoupling oxygen redox. Adv. Mater. 36(26), 2401018 (2024). https://doi.org/10.1002/adma.202401018
- W. Liu, R. Chen, Z. Sang, Z. Li, J. Nie et al., A generalized coordination engineering strategy for single-atom catalysts toward efficient hydrogen peroxide electrosynthesis. Adv. Mater. 36(38), 2406403 (2024). https://doi.org/10.1002/adma.202406403
- M. Liu, H. Su, X. Liu, X. He, P. Tan et al., Dynamic modulation of electron redistribution at the heterogeneous interface nickel hydroxides/platinum boosts acidic oxygen reduction reaction. Nat. Commun. 16, 2826 (2025). https://doi.org/10.1038/s41467-025-58193-8
- Z. Liu, W. Yuan, H. Yang, Z. Kang, M. Ma et al., Phase reconstruction-directed synthesis of oxalate-functionalized nickel hydroxide electrocatalyst for high-yield H2O2 generation at industrial currents. Adv. Funct. Mater. 35(2), 2412198 (2025). https://doi.org/10.1002/adfm.202412198
- H. Ze, X. Chen, X.-T. Wang, Y.-H. Wang, Q.-Q. Chen et al., Molecular insight of the critical role of Ni in Pt-based nanocatalysts for improving the oxygen reduction reaction probed using an in situ SERS borrowing strategy. J. Am. Chem. Soc. 143(3), 1318–1322 (2021). https://doi.org/10.1021/jacs.0c12755
- Q. Zhang, F. Luo, X. Long, X. Yu, K. Qu et al., N, P doped carbon nanotubes confined WN-Ni Mott-Schottky heterogeneous electrocatalyst for water splitting and rechargeable zinc-air batteries. Appl. Catal. B Environ. 298, 120511 (2021). https://doi.org/10.1016/j.apcatb.2021.120511
- B. Cao, G.M. Veith, R.E. Diaz, J. Liu, E.A. Stach et al., Cobalt molybdenum oxynitrides: synthesis, structural characterization, and catalytic activity for the oxygen reduction reaction. Angew. Chem. Int. Ed. 52(41), 10753–10757 (2013). https://doi.org/10.1002/anie.201303197
- Y.-B. Chen, J.-J. Li, Y.-P. Zhu, J. Zou, H. Zhao et al., Vicinal Co atom-coordinated Fe–N–C catalysts to boost the oxygen reduction reaction. J. Mater. Chem. A 10(18), 9886–9891 (2022). https://doi.org/10.1039/D2TA00884J
- Y. Ha, B. Fei, X. Yan, H. Xu, Z. Chen et al., Atomically dispersed Co-pyridinic N-C for superior oxygen reduction reaction. Adv. Energy Mater. 10(46), 2002592 (2020). https://doi.org/10.1002/aenm.202002592
- L. Lin, Y. Ni, L. Shang, H. Sun, Q. Zhang et al., Atomic-level modulation-induced electron redistribution in Co coordination polymers elucidates the oxygen reduction mechanism. ACS Catal. 12(13), 7531–7540 (2022). https://doi.org/10.1021/acscatal.2c01075
- H. Shen, N. Qiu, L. Yang, X. Guo, K. Zhang et al., Boosting oxygen reduction for high-efficiency H2O2 electrosynthesis on oxygen-coordinated Co-N-C catalysts. Small 18(17), 2200730 (2022). https://doi.org/10.1002/smll.202200730
- J. Wang, H. Zhong, J. Yang, H. Li, P. Tang et al., Tuning the atomic configuration environment of MnN4 sites by Co cooperation for efficient oxygen reduction. J. Energy Chem. 82, 547–559 (2023). https://doi.org/10.1016/j.jechem.2023.04.010
- Z. Yu, D. Zhang, Y. Wang, F. Liu, F. She et al., Spin manipulation of heterogeneous molecular electrocatalysts by an integrated magnetic field for efficient oxygen redox reactions. Adv. Mater. 36(45), 2470360 (2024). https://doi.org/10.1002/adma.202470360
- K. Sun, J. Dong, H. Sun, X. Wang, J. Fang et al., Co(CN)3 catalysts with well-defined coordination structure for the oxygen reduction reaction. Nat. Catal. 6(12), 1164–1173 (2023). https://doi.org/10.1038/s41929-023-01047-7
- W. Xu, R. Zeng, M. Rebarchik, A. Posada-Borbón, H. Li et al., Atomically dispersed Zn/Co-N-C as ORR electrocatalysts for alkaline fuel cells. J. Am. Chem. Soc. 146(4), 2593–2603 (2024). https://doi.org/10.1021/jacs.3c11355
- Y. Yang, Y. Xiao, L. Zhang, H.-T. Wang, K.-H. Chen et al., Encaging Co nanop in atomic CoN4-dispersed graphite nanopocket evokes high oxygen reduction activity for flexible Zn-air battery. Appl. Catal. B Environ. Energy 347, 123792 (2024). https://doi.org/10.1016/j.apcatb.2024.123792
- M. Jiang, H. Wang, M. Zhu, X. Luo, Y. He et al., Review on strategies for improving the added value and expanding the scope of CO2 electroreduction products. Chem. Soc. Rev. 53(10), 5149–5189 (2024). https://doi.org/10.1039/D3CS00857F
- D. Karapinar, C.E. Creissen, J.G. Rivera de la Cruz, M.W. Schreiber, M. Fontecave, Electrochemical CO2 reduction to ethanol with copper-based catalysts. ACS Energy Lett. 6(2), 694–706 (2021). https://doi.org/10.1021/acsenergylett.0c02610
- Y. Lin, T. Wang, L. Zhang, G. Zhang, L. Li et al., Tunable CO2 electroreduction to ethanol and ethylene with controllable interfacial wettability. Nat. Commun. 14(1), 3575 (2023). https://doi.org/10.1038/s41467-023-39351-2
- A. Shayesteh Zeraati, F. Li, T. Alkayyali, R. Dorakhan, E. Shirzadi et al., Carbon- and energy-efficient ethanol electrosynthesis via interfacial cation enrichment. Nat Synth 4(1), 75–83 (2025). https://doi.org/10.1038/s44160-024-00662-x
- Z. Jiang, W. Sun, H. Shang, W. Chen, T. Sun et al., Atomic interface effect of a single atom copper catalyst for enhanced oxygen reduction reactions. Energy Environ. Sci. 12(12), 3508–3514 (2019). https://doi.org/10.1039/C9EE02974E
- H. Li, Y. Wen, M. Jiang, Y. Yao, H. Zhou et al., Understanding of neighboring Fe-N4-C and Co-N4-C dual active centers for oxygen reduction reaction. Adv. Funct. Mater. 31(22), 2011289 (2021). https://doi.org/10.1002/adfm.202011289
- L. Zong, K. Fan, W. Wu, L. Cui, L. Zhang et al., Anchoring single copper atoms to microporous carbon spheres as high-performance electrocatalyst for oxygen reduction reaction. Adv. Funct. Mater. 31(41), 2104864 (2021). https://doi.org/10.1002/adfm.202104864
- W. Zhou, B. Li, X. Liu, J. Jiang, S. Bo et al., In situ tuning of platinum 5d valence states for four-electron oxygen reduction. Nat. Commun. 15(1), 6650 (2024). https://doi.org/10.1038/s41467-024-51157-4
- Q. Zhang, P. Kumar, X. Zhu, R. Daiyan, N.M. Bedford et al., Electronically modified atomic sites within a multicomponent Co/Cu composite for efficient oxygen electroreduction. Adv. Energy Mater. 11(17), 2100303 (2021). https://doi.org/10.1002/aenm.202100303
- Z. Liu, Z. Wang, D. Lv, H. Yang, Z. Kang et al., Efficient electrosynthesis of hydrogen peroxide enabled by a hierarchical hollow RE–P–O (RE = Sm, La, Gd) architecture with open channels. Adv. Mater. 37(7), 2311997 (2025). https://doi.org/10.1002/adma.202311997
- L. Yin, M. Sun, S. Zhang, Y. Huang, B. Huang et al., Chlorine axial coordination activated lanthanum single atoms for efficient oxygen electroreduction with maximum utilization. Adv. Mater. 37(7), 2416387 (2025). https://doi.org/10.1002/adma.202416387
- R. Cheng, X. He, M. Jiang, X. Shao, W. Tang et al., F-p-d gradient orbital coupling induced spin state enhancement of atomic Fe sites for efficient and stable oxygen reduction reaction. Adv. Funct. Mater. 35(29), 2425138 (2025). https://doi.org/10.1002/adfm.202425138
- X.-Z. Yue, Y.-C. Liu, B.-A. Lu, X. Du, W. Lei et al., Inherent anti-Fenton property of single-atom rhenium for the ultra-durable oxygen reduction reaction. Energy Environ. Sci. 17(16), 5892–5900 (2024). https://doi.org/10.1039/D4EE02375G
- S. Ozkan, S.J. Kim, D.N. Miller, J.T.S. Irvine, A new approach to fuel cell electrodes: lanthanum aluminate yielding fine Pt nanop exsolution for oxygen reduction reaction. Adv. Energy Mater. 14(15), 2303025 (2024). https://doi.org/10.1002/aenm.202303025
- X. Han, T. Zhang, W. Chen, B. Dong, G. Meng et al., Mn—N4 oxygen reduction electrocatalyst: operando investigation of active sites and high performance in zinc–air battery. Adv. Energy Mater. 11(6), 2002753 (2021). https://doi.org/10.1002/aenm.202002753
- H. Li, G. Yan, H. Zhao, P.C. Howlett, X. Wang et al., Earthworm-inspired Co/Co3O4/CoF2@NSC nanofibrous electrocatalyst with confined channels for enhanced ORR/OER performance. Adv. Mater. 36(26), 2311272 (2024). https://doi.org/10.1002/adma.202311272
- Z. Wang, H. Jin, T. Meng, K. Liao, W. Meng et al., Fe, Cu-coordinated ZIF-derived carbon framework for efficient oxygen reduction reaction and zinc–air batteries. Adv. Funct. Mater. 28(39), 1802596 (2018). https://doi.org/10.1002/adfm.201802596
- Z. Ji, C. Qiu, P. Li, Y. Li, J. Shi et al., Atomic modulation of FeN4 sites on hollow carbon nanospheres by neighboring Mn atoms for ultra-stable Zn-air batteries. Chem. Eng. J. 500, 157505 (2024). https://doi.org/10.1016/j.cej.2024.157505
- L. Qiu, Z. Wu, Y. Liu, Z. Qin, Y. Liu et al., Mn doping at high-activity octahedral vacancies of γ-Fe2O3 for oxygen reduction reaction electrocatalysis in metal-air batteries. Angew. Chem. Int. Ed. 64(12), e202421918 (2025). https://doi.org/10.1002/anie.202421918
- Y. Li, A. Huang, L. Zhou, B. Li, M. Zheng et al., Main-group element-boosted oxygen electrocatalysis of Cu-N-C sites for zinc-air battery with cycling over 5000 h. Nat. Commun. 15(1), 8365 (2024). https://doi.org/10.1038/s41467-024-52494-0
- W. Zhang, B. Feng, L. Huang, Y. Liang, J. Chen et al., Fe/Cu diatomic sites dispersed on nitrogen-doped mesoporous carbon for the boosted oxygen reduction reaction in Mg-air and Zn-air batteries. Appl. Catal. B Environ. Energy 358, 124450 (2024). https://doi.org/10.1016/j.apcatb.2024.124450
- J. Xia, M. Zhou, H. Gao, F. He, Z. Du et al., Regulation of Ni/Co ratio and gas transport in high-order Ruddlesden–Popper perovskite air electrodes for protonic ceramic electrochemical cells. Adv. Funct. Mater. 34(39), 2403493 (2024). https://doi.org/10.1002/adfm.202403493
- Z. Yang, F. Lai, Q. Mao, C. Liu, S. Peng et al., Breaking the mutual-constraint of bifunctional oxygen electrocatalysis via direct O-O coupling on high-valence Ir single-atom on MnOx. Adv. Mater. 37(3), 2412950 (2025). https://doi.org/10.1002/adma.202412950
- J. Li, M. Chen, D.A. Cullen, S. Hwang, M. Wang et al., Atomically dispersed manganese catalysts for oxygen reduction in proton-exchange membrane fuel cells. Nat. Catal. 1(12), 935–945 (2018). https://doi.org/10.1038/s41929-018-0164-8
- L. Zhang, Y. Dong, L. Li, Y. Shi, Y. Zhang et al., Concurrently boosting activity and stability of oxygen reduction reaction catalysts via judiciously crafting Fe-Mn dual atoms for fuel cells. Nano-Micro Lett. 17(1), 88 (2024). https://doi.org/10.1007/s40820-024-01580-5
- J. Bai, T. Zhao, M. Xu, B. Mei, L. Yang et al., Monosymmetric Fe-N4 sites enabling durable proton exchange membrane fuel cell cathode by chemical vapor modification. Nat. Commun. 15(1), 4219 (2024). https://doi.org/10.1038/s41467-024-47817-0
- L. Shi, D. Liu, X. Lin, R. Cheng, F. Liu et al., Stable and high-performance flow H2-O2 fuel cells with coupled acidic oxygen reduction and alkaline hydrogen oxidation reactions. Adv. Mater. 36(23), 2314077 (2024). https://doi.org/10.1002/adma.202314077
- G. Chen, X. Qiu, S. Liu, Y. Cui, Y. Sun et al., Mn–N–C with high-density atomically dispersed Mn active sites for the oxygen reduction reaction. Angew. Chem. Int. Ed. 64(26), e202503934 (2025). https://doi.org/10.1002/anie.202503934
- J. Tian, Y. Song, X. Hao, X. Wang, Y. Shen et al., Greatly enhanced oxygen reduction reaction in anion exchange membrane fuel cell and Zn-air battery via hole inner edge reconstruction of 2D Pd nanomesh. Adv. Mater. 37(1), e2412051 (2025). https://doi.org/10.1002/adma.202412051
- Z. Sang, Y. Qiao, R. Chen, L. Yin, F. Hou et al., Internal hydrogen-bond enhanced two-electron oxygen reduction reaction for π-d conjugated metal-organic framework to H2O2 synthesis. Nat. Commun. 16(1), 4050 (2025). https://doi.org/10.1038/s41467-025-58628-2
- H. Huang, M. Sun, S. Li, S. Zhang, Y. Lee et al., Enhancing H2O2 electrosynthesis at industrial-relevant current in acidic media on diatomic cobalt sites. J. Am. Chem. Soc. 146(13), 9434–9443 (2024). https://doi.org/10.1021/jacs.4c02031
- F. Yang, C. Feng, S. Zuo, Q. Wang, F. Wei et al., Photocatalytic H2O2 production with >30% quantum efficiency via monovalent copper dynamics. J. Am. Chem. Soc. 147(20), 17112–17120 (2025). https://doi.org/10.1021/jacs.5c02450
- H. Gong, Z. Gong, J. Liu, G. Ye, H. Fei, General and ultrafast photothermal synthesis of atomic metal-nitrogen-carbon catalysts for H2O2 electrosynthesis. Adv. Funct. Mater. 34(25), 2316438 (2024). https://doi.org/10.1002/adfm.202316438
- K.-M. Zhao, D.-X. Wu, W.-K. Wu, J.-B. Nie, F.-S. Geng et al., Identifying high-spin hydroxyl-coordinated Fe3+N4 as the active centre for acidic oxygen reduction using molecular model catalysts. Nat. Catal. 8(5), 422–435 (2025). https://doi.org/10.1038/s41929-025-01324-7
References
D.M. Harraz, K.M. Lodaya, B.Y. Tang, Y Surendranath, Homogeneous-heterogeneous bifunctionality in Pd-catalyzed vinyl acetate synthesis. Science 388(6742), eads7913 (2025). https://doi.org/10.1126/science.ads7913
M. Luo, Z. Zhao, Y. Zhang, Y. Sun, Y. Xing et al., PdMo bimetallene for oxygen reduction catalysis. Nature 574(7776), 81–85 (2019). https://doi.org/10.1038/s41586-019-1603-7
Y. Yan, S. Xu, H. Li, N.C.S. Selvam, J.Y. Lee et al., Perpendicularly anchored ReSe2 nanoflakes on reduced graphene oxide support for highly efficient hydrogen evolution reactions. Chem. Eng. J. 405, 126728 (2021). https://doi.org/10.1016/j.cej.2020.126728
R. Chattot, O. Le Bacq, V. Beermann, S. Kühl, J. Herranz et al., Surface distortion as a unifying concept and descriptor in oxygen reduction reaction electrocatalysis. Nat. Mater. 17(9), 827–833 (2018). https://doi.org/10.1038/s41563-018-0133-2
S. Dey, B. Mondal, S. Chatterjee, A. Rana, S. Amanullah et al., Molecular electrocatalysts for the oxygen reduction reaction. Nat. Rev. Chem. 1(12), 98 (2017). https://doi.org/10.1038/s41570-017-0098
T. Fukushima, W. Drisdell, J. Yano, Y. Surendranath, Graphite-conjugated pyrazines as molecularly tunable heterogeneous electrocatalysts. J. Am. Chem. Soc. 137(34), 10926–10929 (2015). https://doi.org/10.1021/jacs.5b06737
A.A. Gewirth, J.A. Varnell, A.M. DiAscro, Nonprecious metal catalysts for oxygen reduction in heterogeneous aqueous systems. Chem. Rev. 118(5), 2313–2339 (2018). https://doi.org/10.1021/acs.chemrev.7b00335
J. Hong, L. Zhang, Q. Zhu, Z. Du, Y. Zhou et al., A macroporous carbon nanoframe for hosting Mott–Schottky Fe–Co/Mo2C sites as an outstanding bi-functional oxygen electrocatalyst. Mater. Horiz. 10(12), 5969–5982 (2023). https://doi.org/10.1039/D3MH01237A
J. Wang, M. Sun, X. Zhang, J. Liu, J. He et al., pH-dependent urea electrooxidation: from mechanism to catalysts and applications. Adv. Mater. 38(4), e15043 (2026). https://doi.org/10.1002/adma.202515043
Z. Hou, C. Cui, Y. Yang, Z. Huang, Y. Zhuang et al., Strong metal-support interactions in heterogeneous oxygen electrocatalysis. Small 20(52), 2407167 (2024). https://doi.org/10.1002/smll.202407167
C. Hu, G. Xing, W. Han, Y. Hao, C. Zhang et al., Inhibiting demetalation of Fe-N-C via Mn sites for efficient oxygen reduction reaction in zinc-air batteries. Adv. Mater. 36(32), 2405763 (2024). https://doi.org/10.1002/adma.202405763
J.J. Huang, Y. Yang, D. Weinstock, C.R. Bundschu, Q. Li et al., Multimodal in situ X-ray mechanistic studies of a bimetallic oxide electrocatalyst in alkaline media. Nat. Catal. 8(2), 116–125 (2025). https://doi.org/10.1038/s41929-025-01289-7
B. Ji, J. Gou, Y. Zheng, X. Pu, Y. Wang et al., Coordination chemistry of large-sized yttrium single-atom catalysts for oxygen reduction reaction. Adv. Mater. 35(24), 2300381 (2023). https://doi.org/10.1002/adma.202300381
N. Kuwamura, T. Konno, Heterometallic coordination polymers as heterogeneous electrocatalysts. Inorg. Chem. Front. 8(10), 2634–2649 (2021). https://doi.org/10.1039/d1qi00112d
Y. Li, H.-R. Wu, Y. Yu, M.-Y. Chen, K.-M. Zhao et al., High-shell sulfur doping enhances Mn-N4 spin states and boosts oxygen reduction reaction performance in both acidic and alkaline media. Small 21(11), 2411678 (2025). https://doi.org/10.1002/smll.202411678
Y. Pei, D.P. Wilkinson, E. Gyenge, Insights into the electrochemical behavior of manganese oxides as catalysts for the oxygen reduction and evolution reactions: monometallic core-shell Mn/Mn3O4. Small 19(19), 2204585 (2023). https://doi.org/10.1002/smll.202204585
S.C. Perry, D. Pangotra, L. Vieira, L.-I. Csepei, V. Sieber et al., Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3(7), 442–458 (2019). https://doi.org/10.1038/s41570-019-0110-6
F.N.I. Sari, Y.-C. Lai, Y.-J. Huang, X.-Y. Wei, H. Pourzolfaghar et al., Electronic structure engineering in NiFe sulfide via a third metal doping as efficient bifunctional OER/ORR electrocatalyst for rechargeable zinc-air battery. Adv. Funct. Mater. 34(21), 2310181 (2024). https://doi.org/10.1002/adfm.202310181
T. Sharifi, E. Gracia-Espino, A. Chen, G. Hu, T. Wågberg, Oxygen reduction reactions on single-or few-atom discrete active sites for heterogeneous catalysis. Adv. Energy Mater. 10(11), 1902084 (2020). https://doi.org/10.1002/aenm.201902084
J. Song, Z.-X. Qian, J. Yang, X.-M. Lin, Q. Xu et al., In situ/operando investigation for heterogeneous electro-catalysts: from model catalysts to state-of-the-art catalysts. ACS Energy Lett. 9(9), 4414–4440 (2024). https://doi.org/10.1021/acsenergylett.4c01488
D.-W. Wang, D. Su, Heterogeneous nanocarbon materials for oxygen reduction reaction. Energy Environ. Sci. 7(2), 576 (2014). https://doi.org/10.1039/c3ee43463j
X. Wang, N. Zhang, H. Shang, H. Duan, Z. Sun et al., Precisely designing asymmetrical selenium-based dual-atom sites for efficient oxygen reduction. Nat. Commun. 16(1), 470 (2025). https://doi.org/10.1038/s41467-025-55862-6
H. Tian, A. Song, H. Tian, J. Liu, G. Shao et al., Single-atom catalysts for high-energy rechargeable batteries. Chem. Sci. 12(22), 7656–7676 (2021). https://doi.org/10.1039/d1sc00716e
Y. Wang, G.I.N. Waterhouse, L. Shang, T. Zhang, Electrocatalytic oxygen reduction to hydrogen peroxide: from homogeneous to heterogeneous electrocatalysis. Adv. Energy Mater. 11(15), 2003323 (2021). https://doi.org/10.1002/aenm.202003323
L.-H. Xu, Q. Wang, L. Hu, D. Shen, S. Chu et al., Engineering asymmetric bimetallic CoM (M = Ni, Fe, Mn, Cu) nanops encapsulated in freestanding wood-derived carbon electrodes for enhanced ORR kinetics in zinc-air batteries. Small 21(5), e2410290 (2025). https://doi.org/10.1002/smll.202410290
Y. Yan, R. Yu, M. Liu, Z. Qu, J. Yang et al., General synthesis of neighboring dual-atomic sites with a specific pre-designed distance via an interfacial-fixing strategy. Nat. Commun. 16(1), 334 (2025). https://doi.org/10.1038/s41467-024-55630-y
H. Yang, N. An, Z. Kang, P.W. Menezes, Z. Chen, Understanding advanced transition metal-based two electron oxygen reduction electrocatalysts from the perspective of phase engineering. Adv. Mater. 36(25), 2400140 (2024). https://doi.org/10.1002/adma.202400140
S. Yang, X. Liu, S. Li, W. Yuan, L. Yang et al., The mechanism of water oxidation using transition metal-based heterogeneous electrocatalysts. Chem. Soc. Rev. 53(11), 5593–5625 (2024). https://doi.org/10.1039/d3cs01031g
Z. Jiang, X. Yang, J. Zhang, J. Yang, B. Sun et al., From conventional two-electron to emerging multi-electron zinc-iodine batteries: advantages, challenges, and future perspectives. Adv. Funct. Mater. 35(50), e11754 (2025). https://doi.org/10.1002/adfm.202511754
H. Zhang, H.-C. Chen, S. Feizpoor, L. Li, X. Zhang et al., Tailoring oxygen reduction reaction kinetics of Fe-N-C catalyst via spin manipulation for efficient zinc-air batteries. Adv. Mater. 36(25), e2400523 (2024). https://doi.org/10.1002/adma.202400523
H. Tian, Y. Lei, B. Sun, C.-C. Yang, C.-L. Chen et al.,P-d orbital hybridization induced by transition metal atom sites for room temperature sodium sulfur batteries. Natl. Sci. Rev. 12(7), nwaf241 (2025). https://doi.org/10.1093/nsr/nwaf241
Z. Ma, A. Song, Z. Liu, Y. Guo, X. Yang et al., Nanoconfined expansion behavior of hollow MnS@Carbon anode with extended lithiation cyclic stability. Adv. Funct. Mater. 33(28), 2301112 (2023). https://doi.org/10.1002/adfm.202301112
S. Zhao, Y. Yang, Z. Tang, Insight into structural evolution, active sites, and stability of heterogeneous electrocatalysts. Angew. Chem. Int. Ed. 61(11), e202110186 (2022). https://doi.org/10.1002/anie.202110186
X. Zhao, Z.H. Levell, S. Yu, Y. Liu, Atomistic understanding of two-dimensional electrocatalysts from first principles. Chem. Rev. 122(12), 10675–10709 (2022). https://doi.org/10.1021/acs.chemrev.1c00981
J. Zheng, D. Meng, J. Guo, X. Liu, L. Zhou et al., Defect engineering for enhanced electrocatalytic oxygen reaction on transition metal oxides: the role of metal defects. Adv. Mater. 36(28), 2405129 (2024). https://doi.org/10.1002/adma.202405129
H.-X. Zhong, Y. Zhang, X.-B. Zhang, Superior oxygen reduction electrocatalyst: hollow porous spinel microsphere. Chem 4(2), 196–198 (2018). https://doi.org/10.1016/j.chempr.2018.01.015
B. Zhou, N. Xu, T. Lu, Y. Wang, S. Lou et al., Dual-carbon assisted oxygen vacancy engineering for optimizing Mn(III) sites to enhance Zn–air battery performances. Adv. Funct. Mater. 35(4), 2414269 (2025). https://doi.org/10.1002/adfm.202414269
H. Zhou, H. Liu, Y. Sun, L. Jiang, J. Xiao et al., Synergistic effect of single atomic Ce sites and CeO2 nanops for boosting oxygen reduction reaction. Adv. Energy Mater. 15(14), 2404689 (2025). https://doi.org/10.1002/aenm.202404689
M. Zhou, J. Guo, R. Lu, J. Li, S. Lee et al., Amorphous metal metaphosphate for oxygen reduction. Interdiscip. Mater. 4(2), 309–320 (2025). https://doi.org/10.1002/idm2.12228
X. Zou, Q. Lu, J. Wu, K. Zhang, M. Tang et al., Screening spinel oxide supports for RuO2 to boost bifunctional electrocatalysts for advanced Zn–air batteries. Adv. Funct. Mater. 34(36), 2401134 (2024). https://doi.org/10.1002/adfm.202401134
G. Gao, Z. Sun, X. Chen, Z. Guang, B. Sun et al., Recent advances in hydrogen production coupled with alternative oxidation reactions. Coord. Chem. Rev. 509, 215777 (2024). https://doi.org/10.1016/j.ccr.2024.215777
G. Gao, C. Xiao, R. Zhang, W. Chen, C. Liu et al., ZIF-67 derivatives in electrocatalysis. Coord. Chem. Rev. 523, 216296 (2025). https://doi.org/10.1016/j.ccr.2024.216296
G. Gao, G. Zhao, G. Zhu, B. Sun, Z. Sun et al., Recent advancements in noble-metal electrocatalysts for alkaline hydrogen evolution reaction. Chin. Chemical Lett. 36(1), 109557 (2025). https://doi.org/10.1016/j.cclet.2024.109557
X. Zhang, J. Lu, J. Liu, M. Sun, G. Zhang et al., Curvature geometry-spin electronics-catalytic dynamics coupling in emerging catalytic engineering. Chem. Soc. Rev. 55(5), 2691–2730 (2026). https://doi.org/10.1039/d5cs01114k
Q. Huang, F. Wang, Z. Sun, B. Zhang, W. Li et al., In situ growth and dynamic transformation of nickel chelate nanoarrays into reactive surface reconstituted heterostructure for overall water splitting. Adv. Funct. Mater. 34(46), 2407407 (2024). https://doi.org/10.1002/adfm.202407407
L. Shang, Y. Ni, Y. Wang, W. Yang, L. Wang et al., Single-nanometer spinel with precise cation distribution for enhanced oxygen reduction. Adv. Mater. 36(50), e2413141 (2024). https://doi.org/10.1002/adma.202413141
H. Tian, H. Tian, S. Wang, S. Chen, F. Zhang et al., High-power lithium-selenium batteries enabled by atomic cobalt electrocatalyst in hollow carbon cathode. Nat. Commun. 11(1), 5025 (2020). https://doi.org/10.1038/s41467-020-18820-y
X. Zhang, Z. Song, B. Liu, B. Liu, J. Liu et al., The interfacial charge change enhanced by Pr0.6 Sm0.4 Co0.8 Mn0.2O3 activated peroxymonosulfate was used for the efficient degradation of tetracycline under the nanoscale domain limiting and distance effect. Mater. Today Sustain. 28, 101044 (2024). https://doi.org/10.1016/j.mtsust.2024.101044
X. Zhang, Z. Song, B. Liu, J. Liu, Y. Huang et al., Ni/Fe bimetallic atom cluster activate PMS to promote 1O2 production to efficiently remove BPA. ChemistrySelect 9(8), e202303088 (2024). https://doi.org/10.1002/slct.202303088
Y. Yan, M. Wu, L. Zhou, W. Chen, L. Han et al., Enhancing electrocatalytic activity through targeted local electrolyte micro-environment. Adv. Funct. Mater. 35(19), 2419328 (2025). https://doi.org/10.1002/adfm.202419328
S. Kong, J. Liu, X. Zhang, J. He, G. Zhang et al., Bridging organic and inorganic domains: advances and applications of hybrid materials in electrocatalysis. Adv. Energy Mater. 16(5), e05010 (2026). https://doi.org/10.1002/aenm.202505010
Z. Song, X. Zhang, B. Liu, J. Liu, Y. Huang et al., Highly active Ni atomic clusters loaded with coal gasification slag derivatives effectively remove tetracycline by activating persulfate to enhance electron transfer ability. J. Water Process Eng. 66, 105982 (2024). https://doi.org/10.1016/j.jwpe.2024.105982
X. Shi, Y. Jiang, B. Zeng, Z. Sun, M. Yun et al., In situ electrochemical production of solid peroxide from urine. Nat. Catal. 8(1), 67–78 (2025). https://doi.org/10.1038/s41929-024-01277-3
J. Liu, Z. Song, B. Liu, X. Zhang, Y. Huang et al., Bimetallic Ni/Fe atom cluster catalysts enhance non-free radical degradation of organic pollutant phenol. Catal. Lett. 154(5), 2182–2196 (2024). https://doi.org/10.1007/s10562-023-04472-2
J. Liu, Y. Huang, X. Song, Z. Song, X. Zhang et al., Ni-Co synergistic regulation of catalyst surface charge density for efficient DNA base degradation in water. J. Water Process. Eng. 71, 107313 (2025). https://doi.org/10.1016/j.jwpe.2025.107313
G. Zhang, X. Zhang, J. Liu, J. He, W. Ge et al., Transition metal-based electrocatalysts for CO2 reduction towards ethanol. Coord. Chem. Rev. 550, 217403 (2026). https://doi.org/10.1016/j.ccr.2025.217403
C. Li, Y. Yang, J. Lu, L. Ren, X. Zhang et al., Micro-Cu doped Co3O4 as an effective oxygen reduction nano-flower-like catalyst to enhance the power output of air cathode microbial fuel cell. Catal. Lett. 154(11), 6080–6093 (2024). https://doi.org/10.1007/s10562-024-04779-8
Y. Huang, Z. Song, B. Liu, X. Zhang, J. Liu et al., Highly active Co is injected into the PrSmMnO3 parent structure to promote the 1O2 pathway to efficiently degrade residual chloroquine phosphate in wastewater. Catal. Lett. 155(1), 3 (2024). https://doi.org/10.1007/s10562-024-04844-2
M. Du, B. Chu, Q. Wang, C. Li, Y. Lu et al., Dual Fe/I single-atom electrocatalyst for high-performance oxygen reduction and wide-temperature quasi-solid-state Zn-air batteries. Adv. Mater. 36(47), 2412978 (2024). https://doi.org/10.1002/adma.202412978
J. Zhang, Y. Mou, W. Suo, S. Yang, J. Shen et al., Single-atomic Co-N-C sites anchored on helical carbonaceous nanotubes for the oxygen reduction reaction. Adv. Funct. Mater. 35(12), 2417621 (2025). https://doi.org/10.1002/adfm.202417621
C. Brea, G. Hu, Dual-atom catalysts for the oxygen reduction reaction: unraveling atomic structures under reaction conditions. J. Am. Chem. Soc. 147(22), 19210–19216 (2025). https://doi.org/10.1021/jacs.5c04776
Y. Cao, Y. Liu, X. Zheng, J. Yang, H. Wang et al., Quantifying asymmetric coordination to correlate with oxygen reduction activity in Fe-based single-atom catalysts. Angew. Chem. Int. Ed. 64(14), e202423556 (2025). https://doi.org/10.1002/anie.202423556
X. Cui, R. Jin, L. Gao, M. Wu, Y. Liu et al., High-loading single atoms via hierarchically porous nanospheres for oxygen reduction reaction with superior activity and durability. Adv. Funct. Mater. 35(39), 2510108 (2025). https://doi.org/10.1002/adfm.202510108
M. Dan, X. Zhang, C. Du, Z. Guo, J. Zhang et al., Atomically dispersed Fe confined into MnO nanoclusters enhances alkaline oxygen reduction activity and stability. Angew. Chem. Int. Ed. 64(29), e202501531 (2025). https://doi.org/10.1002/anie.202501531
M. Deng, D. Wang, Y. Li, General design concept of high-performance single-atom-site catalysts for H2O2 electrosynthesis. Adv. Mater. 36(24), e2314340 (2024). https://doi.org/10.1002/adma.202314340
H. Huang, M. Sun, K. Chen, Y. Che, X. Tang et al., Unlocking the potential of Mn-based catalyst for durable two-electron oxygen reduction in acid at high current densities. Angew. Chem. Int. Ed. 64(33), e202511844 (2025). https://doi.org/10.1002/anie.202511844
S.-M. Jung, S. Kim, J. An, K.-S. Kim, M. Kim et al., Synergistic effects of Co–N4 and Ni–N4 sites in 2D conductive metal–organic framework electrocatalysts for enhanced oxygen reduction reaction performance. ACS Catal. 15(7), 5568–5576 (2025). https://doi.org/10.1021/acscatal.4c05773
S. Lin, J. Wang, J. Chen, P. Lin, H. Wang et al., Electrochemical pilot H2O2 production by solid-state electrolyte reactor: insights from a hybrid catalyst for 2-electron oxygen reduction reaction. Angew. Chem. Int. Ed. 64(19), e202502144 (2025). https://doi.org/10.1002/anie.202502144
C. Liu, W. Shi, Y. Yuan, K. Zhu, Q. Zhang et al., Cascade hydrogen peroxide reduction reaction endows Cu-Fe dual-atom catalyst with durable oxygen reduction performance. Adv. Funct. Mater. 35(35), 2503079 (2025). https://doi.org/10.1002/adfm.202503079
C. Liu, R. Yang, J. Wang, B. Liu, X. Chang et al., Synergistic catalysts with Fe single atoms and Fe3C clusters for accelerated oxygen adsorption kinetics in oxygen reduction reaction. Angew. Chem. Int. Ed. 64(21), e202501266 (2025). https://doi.org/10.1002/anie.202501266
L. Liu, F. Chen, H. Yang, X. Yan, J. Ren et al., Asymmetric coordination strategy of Cu single-atom catalyst for robust all-pH oxygen reduction reaction. Small 21(32), e2503745 (2025). https://doi.org/10.1002/smll.202503745
M. Liu, Y. Li, L. Yang, P. Zhao, J. Li et al., Defect-triggered orbital hybridization in FeMn dual-atom catalysts toward Sabatier-optimized oxygen reduction. Angew. Chem. Int. Ed. 64(28), e202505268 (2025). https://doi.org/10.1002/anie.202505268
S. Liu, Q. Meyer, D. Xu, Y. Cheng, L. Osmieri et al., Breaking the activity and stability trade-off of platinum-free catalysts for the oxygen reduction reaction in hydrogen fuel cells. ACS Nano 19(21), 19524–19551 (2025). https://doi.org/10.1021/acsnano.5c03610
Y. Liu, Y. Yang, X. Lin, Y. Lin, Z. Zhuo et al., The geometric-electronic coupled design of diatomic catalyst towards oxygen reduction reaction. Nat. Commun. 16(1), 5158 (2025). https://doi.org/10.1038/s41467-025-60170-0
X. Lu, F. Xiang, S. Li, W. Liu, Y. Zeng et al., Hydrogen-bond-assisted synthesis of single-atom and nanocluster synergistic sites for enhanced oxygen reduction reaction. Adv. Funct. Mater. 35(40), 2506982 (2025). https://doi.org/10.1002/adfm.202506982
Q. Ma, Y. Liao, Q. Zhao, R. Gan, Y. Ran et al., Triggering synergistic electronic effect via electron-directed transfer within PtNPs-Fe/NC oxygen reduction catalyst for zinc-air batteries. Small 21(14), 2500344 (2025). https://doi.org/10.1002/smll.202500344
D. Shen, F. Sun, Z. Liang, H. Fu, L. Wang, Axial-N induced square-pyramidal crystal filed of atomically iron sites for enhancing acidic oxygen reduction. Angew. Chem. Int. Ed. 64(33), e202505937 (2025). https://doi.org/10.1002/anie.202505937
Z. Wan, Z. Ma, X. Deng, Y. Wu, J. Li et al., Weakening the dissociation barrier of hydroxyl in Fe–N–C catalysts via precisely manipulating d–p orbital hybridization behaviors for efficient oxygen reduction reaction. Adv. Energy Mater. 15(31), 2501630 (2025). https://doi.org/10.1002/aenm.202501630
P. Wang, Y. Xu, Y. Li, P. Xie, H. Li et al., Engineering active CeO2/Fe3C interfacial sites to generate high-charge-density Fe for enhanced oxygen reduction reaction efficiency. Adv. Funct. Mater. 35(43), 2503577 (2025). https://doi.org/10.1002/adfm.202503577
H. Tian, A. Song, P. Zhang, K. Sun, J. Wang et al., High durability of Fe–N–C single-atom catalysts with carbon vacancies toward the oxygen reduction reaction in alkaline media. Adv. Mater. 35(14), 2210714 (2023). https://doi.org/10.1002/adma.202210714
B. Zhang, J. Dang, H. Li, J.-J. Wang, D. Xu et al., Orderly stacked “tile” architecture with single-atom iron boosts oxygen reduction in liquid and solid-state Zn–air batteries. Adv. Funct. Mater. 35(34), 2502834 (2025). https://doi.org/10.1002/adfm.202502834
T. Zhang, W. Wang, W. Liu, Z. Guo, J. Liu, Residual ligand-functionalized ultrathin Ni(OH)2 via reconstruction for high-rate HO2− electrosynthesis. Nat. Commun. 16(1), 5240 (2025). https://doi.org/10.1038/s41467-025-60467-0
Z. Zhang, Z. Zheng, N. Ma, E. Picheau, N. Sakai et al., Composition tuning and heterostructure construction of Fe-doped Co-Ni hydroxide nanosheets for boosting oxygen electrocatalysis in rechargeable Zn-air batteries. Chem. Eng. J. 509, 161248 (2025). https://doi.org/10.1016/j.cej.2025.161248
Y. Zhao, Z. Gao, S. Zhang, X. Guan, W. Xu et al., Asymmetric-charge-distributed Co-Mn diatomic catalyst enables efficient oxygen reduction reaction. Adv. Funct. Mater. 35(37), 2504260 (2025). https://doi.org/10.1002/adfm.202504260
J. Zou, L. Bao, Q. Sun, C. Bao, H. Chen et al., Oxygen reduction reaction catalysts for zinc-air batteries featuring single cobalt atoms in a nitrogen-doped 3D-interconnected porous graphene framework. Small 21(8), 2409506 (2025). https://doi.org/10.1002/smll.202409506
C. Brea, G. Hu, Mechanistic insight into dual-metal-site catalysts for the oxygen reduction reaction. ACS Catal. 13(7), 4992–4999 (2023). https://doi.org/10.1021/acscatal.3c00090
X. Cao, H. Guo, Y. Han, M. Li, C. Shang et al., Sandwiching intermetallic Pt3Fe and ionomer with porous N-doped carbon layers for oxygen reduction reaction. Nat. Commun. 16(1), 2851 (2025). https://doi.org/10.1038/s41467-025-58116-7
T. He, Y. Chen, Q. Liu, B. Lu, X. Song et al., Theory-guided regulation of FeN4 spin state by neighboring Cu atoms for enhanced oxygen reduction electrocatalysis in flexible metal–air batteries. Angew. Chem. Int. Ed. 61(27), e202201007 (2022). https://doi.org/10.1002/anie.202201007
J. Huang, C. Yu, J. Li, W. Xiao, J.B. Zhong et al., Rare-earth lanthanum-nitrogen-carbon enhanced by abundant microspores for efficient oxygen reduction reaction. J. Energy Chem. 106, 812–822 (2025). https://doi.org/10.1016/j.jechem.2025.02.021
M. Tong, F. Sun, Y. Xie, Y. Wang, Y. Yang et al., Operando cooperated catalytic mechanism of atomically dispersed Cu−N4 and Zn−N4 for promoting oxygen reduction reaction. Angew. Chem. Int. Ed. 60(25), 14005–14012 (2021). https://doi.org/10.1002/anie.202102053
M. Zhang, H. Li, J. Chen, F.-X. Ma, L. Zhen et al., Transition metal (Co, Ni, Fe, Cu) single-atom catalysts anchored on 3D nitrogen-doped porous carbon nanosheets as efficient oxygen reduction electrocatalysts for Zn-air battery. Small 18(34), e2202476 (2022). https://doi.org/10.1002/smll.202202476
L. Zong, K. Fan, P. Li, F. Lu, B. Li et al., Promoting oxygen reduction reaction on atomically dispersed Fe sites via establishing hydrogen bonding with the neighboring P atoms. Adv. Energy Mater. 13(5), 2203611 (2023). https://doi.org/10.1002/aenm.202203611
R. Jinnouchi, K. Kodama, T. Hatanaka, Y. Morimoto, First principles based mean field model for oxygen reduction reaction. Phys. Chem. Chem. Phys. 13(47), 21070 (2011). https://doi.org/10.1039/c1cp21349k
V. Viswanathan, H.A. Hansen, J. Rossmeisl, J.K. Nørskov, Unifying the 2e− and 4e− reduction of oxygen on metal surfaces. J. Phys. Chem. Lett. 3(20), 2948–2951 (2012). https://doi.org/10.1021/jz301476w
H. Chen, Z. Gao, N.T. Nguyen, Z. Li, R.-T. Gao et al., Bias-free photoelectrochemical water oxidation coupled with electrochemical oxygen reduction reaction via Fe-based electrodes with long-term operation. Adv. Funct. Mater. 35(14), 2418670 (2025). https://doi.org/10.1002/adfm.202418670
H. Liu, J. Huang, K. Feng, R. Xiong, S. Ma et al., Reconstructing the coordination environment of Fe/Co dual-atom sites towards efficient oxygen electrocatalysis for Zn–air batteries. Angew. Chem. Int. Ed. 64(7), e202419595 (2025). https://doi.org/10.1002/anie.202419595
S. Bhattacharyya, D. Samanta, S. Roy, V.P. Haveri Radhakantha, T.K. Maji, In situ stabilization of Au and co nanops in a redox-active conjugated microporous polymer matrix: facile heterogeneous catalysis and electrocatalytic oxygen reduction reaction activity. ACS Appl. Mater. Interfaces 11(5), 5455–5461 (2019). https://doi.org/10.1021/acsami.8b20610
V. Beermann, M. Gocyla, E. Willinger, S. Rudi, M. Heggen et al., Rh-doped Pt–Ni octahedral nanops: understanding the correlation between elemental distribution, oxygen reduction reaction, and shape stability. Nano Lett. 16(3), 1719–1725 (2016). https://doi.org/10.1021/acs.nanolett.5b04636
Y. Chen, J. Xu, P. He, Y. Qiao, S. Guo et al., Metal-air batteries: progress and perspective. Sci. Bull. 67(23), 2449–2486 (2022). https://doi.org/10.1016/j.scib.2022.11.027
J. Sun, T. Tang, S. Zhang, S. Chen, Y. Duan et al., A dual-atom La2 catalyst for the oxygen reduction reaction. Angew. Chem. Int. Ed. 64(34), e202509063 (2025). https://doi.org/10.1002/anie.202509063
Y. Wang, J. Wu, Q. Zhang, Y. Tan, J. Gao et al., Defect-induced Zn–Co pair active site for high-efficiency electrosynthesis of H2O2. Matter 8(12), 102479 (2025). https://doi.org/10.1016/j.matt.2025.102479
P.H. van Langevelde, K. Ležaić, J.F.J. Coelho, D.G.H. Hetterscheid, F. De Bon, Interplay between the oxygen reduction reaction and atom transfer radical polymerization with molecular Cu-based catalysts in water. ACS Catal. 15(16), 14548–14563 (2025). https://doi.org/10.1021/acscatal.5c04928
Y. Zhao, J. Wan, C. Ling, Y. Wang, H. He et al., Acidic oxygen reduction by single-atom Fe catalysts on curved supports. Nature 644(8077), 668–675 (2025). https://doi.org/10.1038/s41586-025-09364-6
M. Sun, J. Chen, Z. Zhang, Y. Jing, M. Zhao et al., Ferromagnetic ordering outperforms coordination effects in governing oxygen reduction catalysis on high-index nickel single crystals. Angew. Chem. Int. Ed. 64(31), e202504869 (2025). https://doi.org/10.1002/anie.202504869
Z. Lu, Z. Wang, Z. Yang, X. Jin, L. Tong et al., Engineering CoN4 and FeN4 dual sites with adjacent nanoclusters on flexible porous carbon fibers for enhanced electrocatalytic oxygen reduction and evolution. Adv. Funct. Mater. 35(16), 2418489 (2025). https://doi.org/10.1002/adfm.202418489
M.L. Rigsby, D.J. Wasylenko, M.L. Pegis, J.M. Mayer, Medium effects are as important as catalyst design for selectivity in electrocatalytic oxygen reduction by iron–porphyrin complexes. J. Am. Chem. Soc. 137(13), 4296–4299 (2015). https://doi.org/10.1021/jacs.5b00359
H. Wang, L. Cao, Y. Feng, J. Chen, W. Feng et al., Facile synthesis of defect-rich Fe-N-C hybrid from fullerene/ferrotetraphenylporphyrin as efficient oxygen reduction electrocatalyst for Zn-air battery. Chin. Chem. Lett. 34(5), 107601 (2023). https://doi.org/10.1016/j.cclet.2022.06.024
Z. Wang, R. Xu, Q. Ye, X. Jin, Z. Lu et al., Tailoring first coordination sphere of dual-metal atom sites boosts oxygen reduction and evolution activities. Adv. Funct. Mater. 34(28), 2315376 (2024). https://doi.org/10.1002/adfm.202315376
L. Wu, Y. Chen, C. Shao, L. Wang, B. Li, Engineering synergetic Fe-co atomic pairs anchored on porous carbon for enhanced oxygen reduction reaction. Adv. Funct. Mater. 34(48), 2408257 (2024). https://doi.org/10.1002/adfm.202408257
P.-F. Xie, H. Zhong, L. Fang, Z. Lyu, W.-J. Yu et al., Molecular Fe-N4 moieties coupled with atomic Co-N4 sites toward improved oxygen reduction performance. Adv. Funct. Mater. 34(32), 2314554 (2024). https://doi.org/10.1002/adfm.202314554
D. Xue, S. Zhao, B.-A. Lu, Y. Yu, Y. Wei et al., Disentangling the activity-stability trade-off of pyrrolic N-coordinated Fe-N4 catalytic sites for long-life oxygen reduction reaction in acidic medium. Adv. Energy Mater. 14(12), 2303733 (2024). https://doi.org/10.1002/aenm.202303733
H. Yang, H. Wang, S. Ji, V. Linkov, R. Wang, Synergy between isolated-Fe3O4 nanops and CNx layers derived from lysine to improve the catalytic activity for oxygen reduction reaction. Int. J. Hydrog. Energy 39(8), 3739–3745 (2014). https://doi.org/10.1016/j.ijhydene.2013.12.160
S. Yin, H. Yi, M. Liu, J. Yang, S. Yang et al., An in situ exploration of how Fe/N/C oxygen reduction catalysts evolve during synthesis under pyrolytic conditions. Nat. Commun. 15, 6229 (2024). https://doi.org/10.1038/s41467-024-50629-x
P. Zhang, H.-C. Chen, H. Zhu, K. Chen, T. Li et al., Inter-site structural heterogeneity induction of single atom Fe catalysts for robust oxygen reduction. Nat. Commun. 15(1), 2062 (2024). https://doi.org/10.1038/s41467-024-46389-3
S. Ji, Y. Wang, H. Liu, X. Lu, C. Guo et al., Regulating the electronic synergy of asymmetric atomic Fe sites with adjacent defects for boosting activity and durability toward oxygen reduction. Adv. Funct. Mater. 34(29), 2314621 (2024). https://doi.org/10.1002/adfm.202314621
P. Rao, Y. Liu, X. Shi, Y. Yu, Y. Zhou et al., Protection of Fe single-atoms by Fe clusters for chlorine-resistant oxygen reduction reaction. Adv. Funct. Mater. 34(46), 2407121 (2024). https://doi.org/10.1002/adfm.202407121
J. Liu, Y. Liu, B. Nan, D. Wang, C. Allen et al., A two-in-one strategy to simultaneously boost the site density and turnover frequency of Fe−N−C oxygen reduction catalysts. Angew. Chem. Int. Ed. 64(14), e202425196 (2025). https://doi.org/10.1002/anie.202425196
J. Qiao, C. Lu, L. Kong, J. Zhang, Q. Lin et al., Spin engineering of Fe-N-C by axial ligand modulation for enhanced bifunctional oxygen catalysis. Adv. Funct. Mater. 34(51), 2409794 (2024). https://doi.org/10.1002/adfm.202409794
R. Sui, B. Liu, C. Chen, X. Tan, C. He et al., Constructing asymmetric Fe-Nb diatomic sites to enhance ORR activity and durability. J. Am. Chem. Soc. 146(38), 26442–26453 (2024). https://doi.org/10.1021/jacs.4c09642
G. Yang, H. Cai, N. Zhang, B. Wang, C. Liang et al., Regulation of d-orbital electron in Fe-N4 by high-entropy atomic clusters for highly active and durable oxygen reduction reaction. Adv. Funct. Mater. 34(46), 2407775 (2024). https://doi.org/10.1002/adfm.202407775
M. Yuan, Y. Liu, Y. Du, Z. Xiao, H. Li et al., Dual-shelled hollow leafy carbon support with atomically dispersed (N, S)-bridged hydroxy-coordinated asymmetric Fe sites for oxygen reduction. Adv. Funct. Mater. 34(29), 2401484 (2024). https://doi.org/10.1002/adfm.202401484
Y. Zhu, B. Zhang, X. Liu, D.-W. Wang, D.S. Su, Unravelling the structure of electrocatalytically active Fe–N complexes in carbon for the oxygen reduction reaction. Angew. Chem. Int. Ed. 53(40), 10673–10677 (2014). https://doi.org/10.1002/anie.201405314
X. Lin, D. Liu, L. Shi, F. Liu, F. Ye et al., Second-shell coordination environment modulation for MnN4 active sites by oxygen doping to boost oxygen reduction performance. Small 2407146 (2024). https://doi.org/10.1002/smll.202407146
Y. Qin, C. Guo, Z. Ou, C. Xu, Q. Lan et al., Regulating single-atom Mn sites by precisely axial pyridinic-nitrogen coordination to stabilize the oxygen reduction. J. Energy Chem. 80, 542–552 (2023). https://doi.org/10.1016/j.jechem.2023.01.048
K. Kim, G. Kim, T. Jeong, W. Lee, Y. Yang et al., Activating the Mn single atomic center for an efficient actual active site of the oxygen reduction reaction by spin-state regulation. J. Am. Chem. Soc. 146(49), 34033–34042 (2024). https://doi.org/10.1021/jacs.4c13137
Y. Song, C. Han, W. Li, X. Yi, Q. Liao et al., Engineering bimetallic cluster architectures: Harnessing unique “remote synergy effect” between Mn and Y for enhanced electrocatalytic oxygen reduction reaction. eScience 5(3), 100332 (2025). https://doi.org/10.1016/j.esci.2024.100332
T. Stracensky, L. Jiao, Q. Sun, E. Liu, F. Yang et al., Bypassing formation of oxide intermediate via chemical vapor deposition for the synthesis of an Mn-N-C catalyst with improved ORR activity. ACS Catal. 13(22), 14782–14791 (2023). https://doi.org/10.1021/acscatal.3c01982
Z. Luo, J. Xie, J. Cheng, F. Wei, S. Lyu et al., Spin-state manipulation of atomic manganese center by phosphide-support interactions for enhanced oxygen reduction. Adv. Mater. 37(29), 2504585 (2025). https://doi.org/10.1002/adma.202504585
Y. Li, Z. Li, K. Shi, L. Luo, H. Jiang et al., Single-atom Mn catalysts via integration with Mn sub nano-clusters synergistically enhance oxygen reduction reaction. Small 20(22), 2309727 (2024). https://doi.org/10.1002/smll.202309727
H. Tian, L. Zeng, Y. Huang, Z. Ma, G. Meng et al., In situ electrochemical Mn(III)/Mn(IV) generation of Mn(II)O electrocatalysts for high-performance oxygen reduction. Nano-Micro Lett. 12(1), 161 (2020). https://doi.org/10.1007/s40820-020-00500-7
S. Ye, D. Zhang, Z. Ou, L. Zheng, W. Liu et al., Electron donor–acceptor activated single atomic sites for boosting oxygen reduction reaction. Adv. Funct. Mater. 34(40), 2405884 (2024). https://doi.org/10.1002/adfm.202405884
C. Zhao, H. Li, W. Wang, J. Xu, B. Shao et al., Revealing the critical role of the lone pair electrons of Bi3+ in electrocatalytic oxygen reduction reaction on Mn-based BiMn2O5 mullite surface. Adv. Funct. Mater. 35(9), 2416257 (2025). https://doi.org/10.1002/adfm.202416257
Y. Jing, Y. Cheng, L. Wang, Y. Liu, B. Yu et al., MOF-derived Co, Fe, and Ni Co-doped N-enriched hollow carbon as efficient electrocatalyst for oxygen reduction reaction. Chem. Eng. J. 397, 125539 (2020). https://doi.org/10.1016/j.cej.2020.125539
B. Li, H. Nam, J. Zhao, J. Chang, N. Lingappan et al., Nanoreactor of nickel-containing carbon-shells as oxygen reduction catalyst. Adv. Mater. 29(7), 1605083 (2017). https://doi.org/10.1002/adma.201605083
H. Li, S. Dai, Y. Wu, Q. Dong, J. Chen et al., Atomic scaled depth correlation to the oxygen reduction reaction performance of single atom Ni alloy to the NiO2 supported Pd nanocrystal. Adv. Sci. 10(11), 2207109 (2023). https://doi.org/10.1002/advs.202207109
M. Li, H. Zhu, Q. Yuan, T. Li, M. Wang et al., Proximity electronic effect of Ni/Co diatomic sites for synergistic promotion of electrocatalytic oxygen reduction and hydrogen evolution. Adv. Funct. Mater. 33(4), 2210867 (2023). https://doi.org/10.1002/adfm.202210867
Y. Sun, K. Fan, J. Li, L. Wang, Y. Yang et al., Boosting electrochemical oxygen reduction to hydrogen peroxide coupled with organic oxidation. Nat. Commun. 15(1), 6098 (2024). https://doi.org/10.1038/s41467-024-50446-2
Q. Zhang, Z. Zheng, R. Gao, X. Xiao, M. Jiao et al., Constructing bipolar dual-active sites through high-entropy-induced electric dipole transition for decoupling oxygen redox. Adv. Mater. 36(26), 2401018 (2024). https://doi.org/10.1002/adma.202401018
W. Liu, R. Chen, Z. Sang, Z. Li, J. Nie et al., A generalized coordination engineering strategy for single-atom catalysts toward efficient hydrogen peroxide electrosynthesis. Adv. Mater. 36(38), 2406403 (2024). https://doi.org/10.1002/adma.202406403
M. Liu, H. Su, X. Liu, X. He, P. Tan et al., Dynamic modulation of electron redistribution at the heterogeneous interface nickel hydroxides/platinum boosts acidic oxygen reduction reaction. Nat. Commun. 16, 2826 (2025). https://doi.org/10.1038/s41467-025-58193-8
Z. Liu, W. Yuan, H. Yang, Z. Kang, M. Ma et al., Phase reconstruction-directed synthesis of oxalate-functionalized nickel hydroxide electrocatalyst for high-yield H2O2 generation at industrial currents. Adv. Funct. Mater. 35(2), 2412198 (2025). https://doi.org/10.1002/adfm.202412198
H. Ze, X. Chen, X.-T. Wang, Y.-H. Wang, Q.-Q. Chen et al., Molecular insight of the critical role of Ni in Pt-based nanocatalysts for improving the oxygen reduction reaction probed using an in situ SERS borrowing strategy. J. Am. Chem. Soc. 143(3), 1318–1322 (2021). https://doi.org/10.1021/jacs.0c12755
Q. Zhang, F. Luo, X. Long, X. Yu, K. Qu et al., N, P doped carbon nanotubes confined WN-Ni Mott-Schottky heterogeneous electrocatalyst for water splitting and rechargeable zinc-air batteries. Appl. Catal. B Environ. 298, 120511 (2021). https://doi.org/10.1016/j.apcatb.2021.120511
B. Cao, G.M. Veith, R.E. Diaz, J. Liu, E.A. Stach et al., Cobalt molybdenum oxynitrides: synthesis, structural characterization, and catalytic activity for the oxygen reduction reaction. Angew. Chem. Int. Ed. 52(41), 10753–10757 (2013). https://doi.org/10.1002/anie.201303197
Y.-B. Chen, J.-J. Li, Y.-P. Zhu, J. Zou, H. Zhao et al., Vicinal Co atom-coordinated Fe–N–C catalysts to boost the oxygen reduction reaction. J. Mater. Chem. A 10(18), 9886–9891 (2022). https://doi.org/10.1039/D2TA00884J
Y. Ha, B. Fei, X. Yan, H. Xu, Z. Chen et al., Atomically dispersed Co-pyridinic N-C for superior oxygen reduction reaction. Adv. Energy Mater. 10(46), 2002592 (2020). https://doi.org/10.1002/aenm.202002592
L. Lin, Y. Ni, L. Shang, H. Sun, Q. Zhang et al., Atomic-level modulation-induced electron redistribution in Co coordination polymers elucidates the oxygen reduction mechanism. ACS Catal. 12(13), 7531–7540 (2022). https://doi.org/10.1021/acscatal.2c01075
H. Shen, N. Qiu, L. Yang, X. Guo, K. Zhang et al., Boosting oxygen reduction for high-efficiency H2O2 electrosynthesis on oxygen-coordinated Co-N-C catalysts. Small 18(17), 2200730 (2022). https://doi.org/10.1002/smll.202200730
J. Wang, H. Zhong, J. Yang, H. Li, P. Tang et al., Tuning the atomic configuration environment of MnN4 sites by Co cooperation for efficient oxygen reduction. J. Energy Chem. 82, 547–559 (2023). https://doi.org/10.1016/j.jechem.2023.04.010
Z. Yu, D. Zhang, Y. Wang, F. Liu, F. She et al., Spin manipulation of heterogeneous molecular electrocatalysts by an integrated magnetic field for efficient oxygen redox reactions. Adv. Mater. 36(45), 2470360 (2024). https://doi.org/10.1002/adma.202470360
K. Sun, J. Dong, H. Sun, X. Wang, J. Fang et al., Co(CN)3 catalysts with well-defined coordination structure for the oxygen reduction reaction. Nat. Catal. 6(12), 1164–1173 (2023). https://doi.org/10.1038/s41929-023-01047-7
W. Xu, R. Zeng, M. Rebarchik, A. Posada-Borbón, H. Li et al., Atomically dispersed Zn/Co-N-C as ORR electrocatalysts for alkaline fuel cells. J. Am. Chem. Soc. 146(4), 2593–2603 (2024). https://doi.org/10.1021/jacs.3c11355
Y. Yang, Y. Xiao, L. Zhang, H.-T. Wang, K.-H. Chen et al., Encaging Co nanop in atomic CoN4-dispersed graphite nanopocket evokes high oxygen reduction activity for flexible Zn-air battery. Appl. Catal. B Environ. Energy 347, 123792 (2024). https://doi.org/10.1016/j.apcatb.2024.123792
M. Jiang, H. Wang, M. Zhu, X. Luo, Y. He et al., Review on strategies for improving the added value and expanding the scope of CO2 electroreduction products. Chem. Soc. Rev. 53(10), 5149–5189 (2024). https://doi.org/10.1039/D3CS00857F
D. Karapinar, C.E. Creissen, J.G. Rivera de la Cruz, M.W. Schreiber, M. Fontecave, Electrochemical CO2 reduction to ethanol with copper-based catalysts. ACS Energy Lett. 6(2), 694–706 (2021). https://doi.org/10.1021/acsenergylett.0c02610
Y. Lin, T. Wang, L. Zhang, G. Zhang, L. Li et al., Tunable CO2 electroreduction to ethanol and ethylene with controllable interfacial wettability. Nat. Commun. 14(1), 3575 (2023). https://doi.org/10.1038/s41467-023-39351-2
A. Shayesteh Zeraati, F. Li, T. Alkayyali, R. Dorakhan, E. Shirzadi et al., Carbon- and energy-efficient ethanol electrosynthesis via interfacial cation enrichment. Nat Synth 4(1), 75–83 (2025). https://doi.org/10.1038/s44160-024-00662-x
Z. Jiang, W. Sun, H. Shang, W. Chen, T. Sun et al., Atomic interface effect of a single atom copper catalyst for enhanced oxygen reduction reactions. Energy Environ. Sci. 12(12), 3508–3514 (2019). https://doi.org/10.1039/C9EE02974E
H. Li, Y. Wen, M. Jiang, Y. Yao, H. Zhou et al., Understanding of neighboring Fe-N4-C and Co-N4-C dual active centers for oxygen reduction reaction. Adv. Funct. Mater. 31(22), 2011289 (2021). https://doi.org/10.1002/adfm.202011289
L. Zong, K. Fan, W. Wu, L. Cui, L. Zhang et al., Anchoring single copper atoms to microporous carbon spheres as high-performance electrocatalyst for oxygen reduction reaction. Adv. Funct. Mater. 31(41), 2104864 (2021). https://doi.org/10.1002/adfm.202104864
W. Zhou, B. Li, X. Liu, J. Jiang, S. Bo et al., In situ tuning of platinum 5d valence states for four-electron oxygen reduction. Nat. Commun. 15(1), 6650 (2024). https://doi.org/10.1038/s41467-024-51157-4
Q. Zhang, P. Kumar, X. Zhu, R. Daiyan, N.M. Bedford et al., Electronically modified atomic sites within a multicomponent Co/Cu composite for efficient oxygen electroreduction. Adv. Energy Mater. 11(17), 2100303 (2021). https://doi.org/10.1002/aenm.202100303
Z. Liu, Z. Wang, D. Lv, H. Yang, Z. Kang et al., Efficient electrosynthesis of hydrogen peroxide enabled by a hierarchical hollow RE–P–O (RE = Sm, La, Gd) architecture with open channels. Adv. Mater. 37(7), 2311997 (2025). https://doi.org/10.1002/adma.202311997
L. Yin, M. Sun, S. Zhang, Y. Huang, B. Huang et al., Chlorine axial coordination activated lanthanum single atoms for efficient oxygen electroreduction with maximum utilization. Adv. Mater. 37(7), 2416387 (2025). https://doi.org/10.1002/adma.202416387
R. Cheng, X. He, M. Jiang, X. Shao, W. Tang et al., F-p-d gradient orbital coupling induced spin state enhancement of atomic Fe sites for efficient and stable oxygen reduction reaction. Adv. Funct. Mater. 35(29), 2425138 (2025). https://doi.org/10.1002/adfm.202425138
X.-Z. Yue, Y.-C. Liu, B.-A. Lu, X. Du, W. Lei et al., Inherent anti-Fenton property of single-atom rhenium for the ultra-durable oxygen reduction reaction. Energy Environ. Sci. 17(16), 5892–5900 (2024). https://doi.org/10.1039/D4EE02375G
S. Ozkan, S.J. Kim, D.N. Miller, J.T.S. Irvine, A new approach to fuel cell electrodes: lanthanum aluminate yielding fine Pt nanop exsolution for oxygen reduction reaction. Adv. Energy Mater. 14(15), 2303025 (2024). https://doi.org/10.1002/aenm.202303025
X. Han, T. Zhang, W. Chen, B. Dong, G. Meng et al., Mn—N4 oxygen reduction electrocatalyst: operando investigation of active sites and high performance in zinc–air battery. Adv. Energy Mater. 11(6), 2002753 (2021). https://doi.org/10.1002/aenm.202002753
H. Li, G. Yan, H. Zhao, P.C. Howlett, X. Wang et al., Earthworm-inspired Co/Co3O4/CoF2@NSC nanofibrous electrocatalyst with confined channels for enhanced ORR/OER performance. Adv. Mater. 36(26), 2311272 (2024). https://doi.org/10.1002/adma.202311272
Z. Wang, H. Jin, T. Meng, K. Liao, W. Meng et al., Fe, Cu-coordinated ZIF-derived carbon framework for efficient oxygen reduction reaction and zinc–air batteries. Adv. Funct. Mater. 28(39), 1802596 (2018). https://doi.org/10.1002/adfm.201802596
Z. Ji, C. Qiu, P. Li, Y. Li, J. Shi et al., Atomic modulation of FeN4 sites on hollow carbon nanospheres by neighboring Mn atoms for ultra-stable Zn-air batteries. Chem. Eng. J. 500, 157505 (2024). https://doi.org/10.1016/j.cej.2024.157505
L. Qiu, Z. Wu, Y. Liu, Z. Qin, Y. Liu et al., Mn doping at high-activity octahedral vacancies of γ-Fe2O3 for oxygen reduction reaction electrocatalysis in metal-air batteries. Angew. Chem. Int. Ed. 64(12), e202421918 (2025). https://doi.org/10.1002/anie.202421918
Y. Li, A. Huang, L. Zhou, B. Li, M. Zheng et al., Main-group element-boosted oxygen electrocatalysis of Cu-N-C sites for zinc-air battery with cycling over 5000 h. Nat. Commun. 15(1), 8365 (2024). https://doi.org/10.1038/s41467-024-52494-0
W. Zhang, B. Feng, L. Huang, Y. Liang, J. Chen et al., Fe/Cu diatomic sites dispersed on nitrogen-doped mesoporous carbon for the boosted oxygen reduction reaction in Mg-air and Zn-air batteries. Appl. Catal. B Environ. Energy 358, 124450 (2024). https://doi.org/10.1016/j.apcatb.2024.124450
J. Xia, M. Zhou, H. Gao, F. He, Z. Du et al., Regulation of Ni/Co ratio and gas transport in high-order Ruddlesden–Popper perovskite air electrodes for protonic ceramic electrochemical cells. Adv. Funct. Mater. 34(39), 2403493 (2024). https://doi.org/10.1002/adfm.202403493
Z. Yang, F. Lai, Q. Mao, C. Liu, S. Peng et al., Breaking the mutual-constraint of bifunctional oxygen electrocatalysis via direct O-O coupling on high-valence Ir single-atom on MnOx. Adv. Mater. 37(3), 2412950 (2025). https://doi.org/10.1002/adma.202412950
J. Li, M. Chen, D.A. Cullen, S. Hwang, M. Wang et al., Atomically dispersed manganese catalysts for oxygen reduction in proton-exchange membrane fuel cells. Nat. Catal. 1(12), 935–945 (2018). https://doi.org/10.1038/s41929-018-0164-8
L. Zhang, Y. Dong, L. Li, Y. Shi, Y. Zhang et al., Concurrently boosting activity and stability of oxygen reduction reaction catalysts via judiciously crafting Fe-Mn dual atoms for fuel cells. Nano-Micro Lett. 17(1), 88 (2024). https://doi.org/10.1007/s40820-024-01580-5
J. Bai, T. Zhao, M. Xu, B. Mei, L. Yang et al., Monosymmetric Fe-N4 sites enabling durable proton exchange membrane fuel cell cathode by chemical vapor modification. Nat. Commun. 15(1), 4219 (2024). https://doi.org/10.1038/s41467-024-47817-0
L. Shi, D. Liu, X. Lin, R. Cheng, F. Liu et al., Stable and high-performance flow H2-O2 fuel cells with coupled acidic oxygen reduction and alkaline hydrogen oxidation reactions. Adv. Mater. 36(23), 2314077 (2024). https://doi.org/10.1002/adma.202314077
G. Chen, X. Qiu, S. Liu, Y. Cui, Y. Sun et al., Mn–N–C with high-density atomically dispersed Mn active sites for the oxygen reduction reaction. Angew. Chem. Int. Ed. 64(26), e202503934 (2025). https://doi.org/10.1002/anie.202503934
J. Tian, Y. Song, X. Hao, X. Wang, Y. Shen et al., Greatly enhanced oxygen reduction reaction in anion exchange membrane fuel cell and Zn-air battery via hole inner edge reconstruction of 2D Pd nanomesh. Adv. Mater. 37(1), e2412051 (2025). https://doi.org/10.1002/adma.202412051
Z. Sang, Y. Qiao, R. Chen, L. Yin, F. Hou et al., Internal hydrogen-bond enhanced two-electron oxygen reduction reaction for π-d conjugated metal-organic framework to H2O2 synthesis. Nat. Commun. 16(1), 4050 (2025). https://doi.org/10.1038/s41467-025-58628-2
H. Huang, M. Sun, S. Li, S. Zhang, Y. Lee et al., Enhancing H2O2 electrosynthesis at industrial-relevant current in acidic media on diatomic cobalt sites. J. Am. Chem. Soc. 146(13), 9434–9443 (2024). https://doi.org/10.1021/jacs.4c02031
F. Yang, C. Feng, S. Zuo, Q. Wang, F. Wei et al., Photocatalytic H2O2 production with >30% quantum efficiency via monovalent copper dynamics. J. Am. Chem. Soc. 147(20), 17112–17120 (2025). https://doi.org/10.1021/jacs.5c02450
H. Gong, Z. Gong, J. Liu, G. Ye, H. Fei, General and ultrafast photothermal synthesis of atomic metal-nitrogen-carbon catalysts for H2O2 electrosynthesis. Adv. Funct. Mater. 34(25), 2316438 (2024). https://doi.org/10.1002/adfm.202316438
K.-M. Zhao, D.-X. Wu, W.-K. Wu, J.-B. Nie, F.-S. Geng et al., Identifying high-spin hydroxyl-coordinated Fe3+N4 as the active centre for acidic oxygen reduction using molecular model catalysts. Nat. Catal. 8(5), 422–435 (2025). https://doi.org/10.1038/s41929-025-01324-7